Therapeutic efficacy of a human papillomavirus type 16 E7 bacterial exotoxin fusion protein adjuvanted with CpG or GPI-0100 in a preclinical mouse model for HPV-associated disease
- PMID: 31010714
- PMCID: PMC6586561
- DOI: 10.1016/j.vaccine.2019.04.043
Therapeutic efficacy of a human papillomavirus type 16 E7 bacterial exotoxin fusion protein adjuvanted with CpG or GPI-0100 in a preclinical mouse model for HPV-associated disease
Abstract
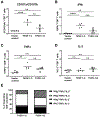
Persistent human papillomavirus (HPV) infection is causally linked to the development of several human cancers, including cervical, vulvar, vaginal, anal, penile, and oropharyngeal cancers. To address the need for a therapeutic vaccine against HPV-associated diseases, here we test and compare the immunogenicity and therapeutic efficacy of a bacterial exotoxin fusion protein covalently linked to the HPV16 E7 oncoprotein adjuvanted with CpG or GPI-0100 in the C3.43 preclinical HPV16-transformed tumor model. We show that TVGV-1 protein vaccine adjuvanted with either CpG or GPI-0100 adjuvant induces a high frequency of E7-specific CD8+ T cells, and both adjuvants are able to assist the immune response in inducing polyfunctional cytokine-secreting lytic T cells that show therapeutic efficacy against well-established C3.43 tumors. CpG-adjuvanted TVGV-1 resulted in higher frequencies of IFNγ secreting and degranulating E7-specific T cells compared to GPI-0100-adjuvanted TVGV-1, resulting in marginally increased in vivo efficacy. Despite minor differences in immune response outcomes, we consider both CpG ODN and GPI-0100 to be promising vaccine adjuvants to increase the immunogenicity and therapeutic efficacy of the TVGV-1 protein for HPV16-driven cancers.
Keywords: CpG adjuvant; GPI-0100 adjuvant; HPV16-induced tumors; Human papillomavirus; Therapeutic vaccine.
Copyright © 2019 The Author(s). Published by Elsevier Ltd.. All rights reserved.
Conflict of interest statement
Disclosure
DMD, JGS, ECJ, KPL, and WMK have no competing interests to declare. JMW, CMW and KH are employees of TheVax Genetics Vaccine Co., Ltd. The funder, TheVax Genetics Vaccine Co., Ltd, played a role in study design and decision to submit article for publication.
Figures
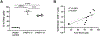


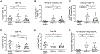
Similar articles
-
A novel recombinant protein vaccine containing the different E7 proteins of the HPV16, 18, 6, 11 E7 linked to the HIV-1 Tat (47-57) improve cytotoxic immune responses.Biotechnol Lett. 2021 Sep;43(9):1933-1944. doi: 10.1007/s10529-021-03166-2. Epub 2021 Jul 27. Biotechnol Lett. 2021. PMID: 34313864
-
Augmented immunogenicity of the HPV16 DNA vaccine via dual adjuvant approach: integration of CpG ODN into plasmid backbone and co-administration with IL-28B gene adjuvant.Virol J. 2025 Jan 8;22(1):3. doi: 10.1186/s12985-024-02604-7. Virol J. 2025. PMID: 39780219 Free PMC article.
-
A novel therapeutic vaccine composed of a rearranged human papillomavirus type 16 E6/E7 fusion protein and Fms-like tyrosine kinase-3 ligand induces CD8+ T cell responses and antitumor effect.Vaccine. 2017 Nov 7;35(47):6459-6467. doi: 10.1016/j.vaccine.2017.09.003. Epub 2017 Oct 10. Vaccine. 2017. PMID: 29029939
-
TriVax-HPV: an improved peptide-based therapeutic vaccination strategy against human papillomavirus-induced cancers.Cancer Immunol Immunother. 2012 Aug;61(8):1307-17. doi: 10.1007/s00262-012-1259-8. Epub 2012 Apr 22. Cancer Immunol Immunother. 2012. PMID: 22527249 Free PMC article. Review.
-
The Use of Both Therapeutic and Prophylactic Vaccines in the Therapy of Papillomavirus Disease.Front Immunol. 2020 Feb 18;11:188. doi: 10.3389/fimmu.2020.00188. eCollection 2020. Front Immunol. 2020. PMID: 32133000 Free PMC article. Review.
Cited by
-
Therapeutic Vaccines for HPV-Associated Malignancies.Immunotargets Ther. 2020 Oct 7;9:167-200. doi: 10.2147/ITT.S273327. eCollection 2020. Immunotargets Ther. 2020. PMID: 33117742 Free PMC article. Review.
-
The association of cervicovaginal Langerhans cells with clearance of human papillomavirus.Front Immunol. 2022 Oct 12;13:918190. doi: 10.3389/fimmu.2022.918190. eCollection 2022. Front Immunol. 2022. PMID: 36311788 Free PMC article. Review.
-
A comprehensive bibliometric analysis (2000-2022) on the mapping of knowledge regarding immunotherapeutic treatments for advanced, recurrent, or metastatic cervical cancer.Front Pharmacol. 2024 May 10;15:1351363. doi: 10.3389/fphar.2024.1351363. eCollection 2024. Front Pharmacol. 2024. PMID: 38799160 Free PMC article.
-
Immune Environment and Immunotherapy in Endometrial Carcinoma and Cervical Tumors.Cancers (Basel). 2023 Mar 29;15(7):2042. doi: 10.3390/cancers15072042. Cancers (Basel). 2023. PMID: 37046702 Free PMC article. Review.
-
Roles of human papillomavirus in cancers: oncogenic mechanisms and clinical use.Signal Transduct Target Ther. 2025 Jan 24;10(1):44. doi: 10.1038/s41392-024-02083-w. Signal Transduct Target Ther. 2025. PMID: 39856040 Free PMC article. Review.
References
-
- Forman D, de Martel C, Lacey CJ, Soerjomataram I, Lortet-Tieulent J, Bruni L, et al. Global burden of human papillomavirus and related diseases. Vaccine. 2012;30 Suppl 5:F12–23. - PubMed
-
- de Sanjose S, Quint WG, Alemany L, Geraets DT, Klaustermeier JE, Lloveras B, et al. Human papillomavirus genotype attribution in invasive cervical cancer: a retrospective cross-sectional worldwide study. Lancet Oncol. 2010;11:1048–56. - PubMed
-
- Serrano B, de Sanjose S, Tous S, Quiros B, Munoz N, Bosch X, et al. Human papillomavirus genotype attribution for HPVs 6, 11, 16, 18, 31, 33, 45, 52 and 58 in female anogenital lesions. Eur J Cancer. 2015;51:1732–41. - PubMed
-
- Kreimer AR, Clifford GM, Boyle P, Franceschi S. Human papillomavirus types in head and neck squamous cell carcinomas worldwide: a systematic review. Cancer Epidemiol Biomarkers Prev. 2005;14:467–75. - PubMed
-
- Doorbar J, Quint W, Banks L, Bravo IG, Stoler M, Broker TR, et al. The biology and life-cycle of human papillomaviruses. Vaccine. 2012;30 Suppl 5:F55–70. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

