Pharmacokinetics and Biodistribution of Pegylated Methotrexate after IV Administration to Mice
- PMID: 31011346
- PMCID: PMC6447882
Pharmacokinetics and Biodistribution of Pegylated Methotrexate after IV Administration to Mice
Abstract
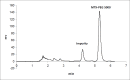
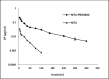
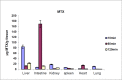
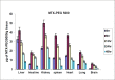
The efficacy of methotrexate (MTX) as an antimetabolite chemotherapeutic agent highly depends on its blood circulation half-life. In our previous study, different conjugates of MTX (MTX-PEG) were synthesized, their physicochemical properties were investigated and MTX-PEG5000 was finally selected as optimum drug-conjugate for further investigations. In the current work, first the stability of MTX-PEG5000 was studied at 37 °C and the results indicated its high stability in plasma (T1/2 = 144 h) and a relatively rapid degradation in tissue homogenate (T1/2 = 24 h). The study of protein binding pointed out that the conjugate was highly protein-bound (95%). The results of pharmacokinetic studies in mice indicated that MTX-PEG5000 had longer plasma distribution and elimination half-lives compared to free MTX (T1/2 α 9.16 min for MTX-PEG5000 versus 2.45 min for MTX and T1/2 β 88.44 for MTX-PEG5000 versus 24.33 min for MTX). Pharmacokinetic parameters also showed higher area under the curve (AUC) of conjugate compared to parent drug (12.33 mg.mL-1.min for MTX-PEG5000 versus 2.64 mg.mL-1.min for MTX). The biodistribution studies demonstrated that MTX-PEG5000 did not highly accumulate in liver and intestine and had a mild and balanced distribution to other organs. Also, the conjugate was measurable in tissues up to 48 h after injection and was detected in the brain, suggesting the possibility of delivering drug to brain tumors.
Keywords: Biodistribution; Methotrexate; Mice; Pegylation; Pharmacokinetics; Stability.
Figures

Similar articles
-
The pharmacokinetics of methotrexate after intravenous administration of methotrexate-loaded proliposomes to rats.Biopharm Drug Dispos. 1994 Jul;15(5):391-407. doi: 10.1002/bdd.2510150506. Biopharm Drug Dispos. 1994. PMID: 7981428
-
Pharmacokinetics of methotrexate after intravenous and intramuscular injection of methotrexate-bearing positively charged liposomes to rats.Biopharm Drug Dispos. 1995 May;16(4):279-93. doi: 10.1002/bdd.2510160404. Biopharm Drug Dispos. 1995. PMID: 7548777
-
Polyethylene glycol conjugates of methotrexate varying in their molecular weight from MW 750 to MW 40000: synthesis, characterization, and structure-activity relationships in vitro and in vivo.Bioconjug Chem. 2002 Jul-Aug;13(4):773-85. doi: 10.1021/bc010098m. Bioconjug Chem. 2002. PMID: 12121133
-
Laser-assisted delivery of topical methotrexate - in vitro investigations.Dan Med J. 2016 Jun;63(6):B5254. Dan Med J. 2016. PMID: 27264947 Review.
-
[Pharmacokinetics of methotrexate in rheumatoid arthritis: therapeutic implications].Therapie. 1997 Mar-Apr;52(2):129-32. Therapie. 1997. PMID: 9231507 Review. French.
Cited by
-
Self-therapeutic metal-based nanoparticles for treating inflammatory diseases.Acta Pharm Sin B. 2023 May;13(5):1847-1865. doi: 10.1016/j.apsb.2022.07.009. Epub 2022 Jul 19. Acta Pharm Sin B. 2023. PMID: 37250153 Free PMC article. Review.
-
DHFR Inhibitors Display a Pleiotropic Anti-Viral Activity against SARS-CoV-2: Insights into the Mechanisms of Action.Viruses. 2023 May 9;15(5):1128. doi: 10.3390/v15051128. Viruses. 2023. PMID: 37243214 Free PMC article.
-
Ceria-based nanotheranostic agent for rheumatoid arthritis.Theranostics. 2020 Oct 25;10(26):11863-11880. doi: 10.7150/thno.49069. eCollection 2020. Theranostics. 2020. PMID: 33204316 Free PMC article.
-
A review of existing strategies for designing long-acting parenteral formulations: Focus on underlying mechanisms, and future perspectives.Acta Pharm Sin B. 2021 Aug;11(8):2396-2415. doi: 10.1016/j.apsb.2021.05.002. Epub 2021 Jun 17. Acta Pharm Sin B. 2021. PMID: 34522592 Free PMC article. Review.
References
-
- Kumar S, Noronha V, Chu E. Antimetabolites. In: DeVita VT, Hellman S, Rosenberg SA, editors. Cancer: Principles and Practice of Oncology. 7th ed. NewYork : Lippincott Williams and Wilkins; 2005.
-
- Chabner B A, Ryan D P, Paz-Ares L, Garcia-Carbonero R, Calabresi P. Antineoplastic Agents. In: Hardmann JG, Limbird LE, Gilman AG, editors. Goodman and Gilman’s the Pharmacological Basis of Therapeutics. New York : McGraw-Hill; 2001. pp. 1399–04.
-
- Crom WR, Evan WE. Methotrexate. In: Evans WE, Schentag JJ, Jusko WJ, editors. Applied pharmacokinetics: principles of therapeutic drug monitoring. 3rd ed. New York: Lippincott Williams and Wilkins; 1992.
-
- Pui CH. Childhood leukemias. N. Engl. J. Med. 1995;332:1618–30. - PubMed
LinkOut - more resources
Full Text Sources
