Retreatment with carboplatin and paclitaxel for recurrent endometrial cancer: A retrospective study of the Memorial Sloan Kettering Cancer Center experience
- PMID: 31011610
- PMCID: PMC6461565
- DOI: 10.1016/j.gore.2019.04.002
Retreatment with carboplatin and paclitaxel for recurrent endometrial cancer: A retrospective study of the Memorial Sloan Kettering Cancer Center experience
Abstract
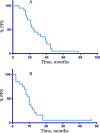
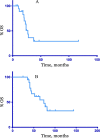
Women with endometrial cancer (EC) frequently receive adjuvant paclitaxel and carboplatin (PC) chemotherapy. There is no standard first line chemotherapy at disease recurrence. Data extrapolated from ovarian cancer has suggested that patients with recurrent EC may benefit from further platinum-based chemotherapy. We performed a retrospective analysis of patients who were retreated with PC chemotherapy for recurrent EC at Memorial Sloan Kettering Cancer Center between January 2000 and December 2014. The median progression free survival (PFS) and overall survival (OS) were estimated using the Kaplan Meier method. Twenty patients were included in the analysis. Patients were re-treated with PC a median of 25 (8-79) months from their original PC. There were no complete responses, 10 (50%) patients had partial response (PR), 3 (15%) had stable disease, 2 (10%) had progression at best response and 5 (20%) were not evaluable by RECIST. A median of 6 cycles of PC were administered (2-9). Four patients (20%) transitioned to paclitaxel only due to carboplatin allergy. At the data cut off, one patient continued PC, and another was off therapy with PR. The remainder (N = 18, 90%) received a median of 2.5 (1-6) further lines of treatments. Median PFS and OS from re-treatment were 10 and 27 months respectively. Median OS from original diagnosis was 74 months. In this small retrospective study, selected patients with recurrent EC who are >6 months from completion of PC derive benefit from retreatment with PC with a response rate of 50%.
Keywords: Carboplatin; Endometrial cancer; Paclitaxel; Retreatment.
Figures
Similar articles
-
Treatment of recurrent ovarian cancer: a retrospective analysis of women treated with single-agent carboplatin originally treated with carboplatin and paclitaxel. The Memorial Sloan-Kettering Cancer Center experience.Gynecol Oncol. 2003 Dec;91(3):584-90. doi: 10.1016/j.ygyno.2003.08.017. Gynecol Oncol. 2003. PMID: 14675681
-
Optimal chemotherapy treatment for women with recurrent ovarian cancer.Curr Oncol. 2007 Oct;14(5):195-208. doi: 10.3747/co.2007.148. Curr Oncol. 2007. PMID: 17938703 Free PMC article.
-
Salvage chemotherapy in recurrent platinum-resistant or refractory epithelial ovarian cancer with Carboplatin and distearoylphosphatidylcholine pegylated liposomal Doxorubicin (lipo-dox®).Asian Pac J Cancer Prev. 2013;14(3):2131-5. doi: 10.7314/apjcp.2013.14.3.2131. Asian Pac J Cancer Prev. 2013. PMID: 23679331
-
Dose-dense chemotherapy with weekly paclitaxel and 3-weekly carboplatin for recurrent ovarian cancer.Taiwan J Obstet Gynecol. 2020 Jan;59(1):21-27. doi: 10.1016/j.tjog.2019.10.003. Taiwan J Obstet Gynecol. 2020. PMID: 32039795 Review.
-
Platinum-based adjuvant chemotherapy on moderate- and high-risk stage I and II epithelian ovarian cancer patients. Long-term single institution experience and literature review.Clin Transl Oncol. 2011 Feb;13(2):121-32. doi: 10.1007/s12094-011-0629-6. Clin Transl Oncol. 2011. PMID: 21324801 Review.
Cited by
-
The Peptide-Drug Conjugate TH1902: A New Sortilin Receptor-Mediated Cancer Therapeutic against Ovarian and Endometrial Cancers.Cancers (Basel). 2022 Apr 8;14(8):1877. doi: 10.3390/cancers14081877. Cancers (Basel). 2022. PMID: 35454785 Free PMC article.
-
Pan-Asian adapted ESMO Clinical Practice Guidelines for the diagnosis, treatment and follow-up of patients with endometrial cancer.ESMO Open. 2023 Feb;8(1):100774. doi: 10.1016/j.esmoop.2022.100774. Epub 2023 Jan 23. ESMO Open. 2023. PMID: 36696825 Free PMC article.
-
Combined PI3K Inhibitor and Eribulin Enhances Anti-Tumor Activity in Preclinical Models of Paclitaxel-Resistant, PIK3CA-Mutated Endometrial Cancer.Cancers (Basel). 2023 Oct 8;15(19):4887. doi: 10.3390/cancers15194887. Cancers (Basel). 2023. PMID: 37835582 Free PMC article.
-
ESGO/ESTRO/ESP Guidelines for the management of patients with endometrial carcinoma.Virchows Arch. 2021 Feb;478(2):153-190. doi: 10.1007/s00428-020-03007-z. Virchows Arch. 2021. PMID: 33604759
-
Looking beyond carboplatin and paclitaxel for the treatment of advanced/recurrent endometrial cancer.Gynecol Oncol. 2022 Dec;167(3):540-546. doi: 10.1016/j.ygyno.2022.10.012. Epub 2022 Oct 22. Gynecol Oncol. 2022. PMID: 36280455 Free PMC article. Review.
References
-
- Alvarez E.A., Brady W.E., Walker J.L. Phase II trial of combination bevacizumab and temsirolimus in the treatment of recurrent or persistent endometrial carcinoma: a gynecologic oncology group study. Gynecol. Oncol. 2013;129:22–27. - PubMed
-
- Diaz L.A., Marabelle A., Delord J.-P. Pembrolizumab therapy for microsatellite instability high (MSI-H) colorectal cancer (CRC) and non-CRC. J. Clin. Oncol. 2017;35 (3071–3071)
-
- Eisenhauer E.A., Therasse P., Bogaerts J. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1) Eur. J. Cancer. 2009;45:228–247. - PubMed
-
- Fleming G.F. Second-line therapy for endometrial cancer: the need for better options. J. Clin. Oncol. 2015;33:3535–3540. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
