Adenovirus-associated antibodies in UK cohort of hemophilia patients: A seroprevalence study of the presence of adenovirus-associated virus vector-serotypes AAV5 and AAV8 neutralizing activity and antibodies in patients with hemophilia A
- PMID: 31011710
- PMCID: PMC6462753
- DOI: 10.1002/rth2.12177
Adenovirus-associated antibodies in UK cohort of hemophilia patients: A seroprevalence study of the presence of adenovirus-associated virus vector-serotypes AAV5 and AAV8 neutralizing activity and antibodies in patients with hemophilia A
Abstract
Background: Current treatment for severe hemophilia A is replacement of deficient factor. Although replacement therapy has improved life expectancy and quality, limitations include frequent infusions and high costs. Gene therapy is a potential alternative that utilizes an adeno-associated virus (AAV) vector containing the human genetic code for factor 8 (FVIII) that transduces the liver, enabling endogenous production of FVIII. Individuals with preexisting immunity to AAV serotypes may be less likely to benefit from this treatment.
Objectives: This study measured seroprevalence of antibodies to AAV5 and 8 in an UK adult hemophilia A cohort.
Patients/methods: Patients were recruited from seven hemophilia centres in the UK. Citrated plasma samples from 100 patients were tested for preexisting activities against AAV5 and 8 using AAV transduction inhibition and total antibodies assays.
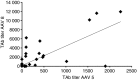
Results: Twent-one percent of patients had antibodies against AAV5 and 23% had antibodies against AAV8. Twenty-five percent and 38% of patients exhibited inhibitors of AAV5 or AAV8 cellular transduction respectively. Overall seroprevalence using either assay against AAV5 was 30% and against AAV8 was 40% in this cohort of hemophilia A patients. Seropositivity for both AAV5 and AAV8 was seen in 24% of participants.
Conclusions: Screening for preexisting immunity may be important in identifying patients most likely to benefit from gene therapy. Clinical studies may be needed to evaluate the impact of preexisting immunity on the safety and efficacy of AAV mediated gene therapy.
Keywords: adeno‐associated viral vectors; hemophilia A; seroprevalence.
Figures
Similar articles
-
Global Seroprevalence of Pre-existing Immunity Against AAV5 and Other AAV Serotypes in People with Hemophilia A.Hum Gene Ther. 2022 Apr;33(7-8):432-441. doi: 10.1089/hum.2021.287. Epub 2022 Mar 16. Hum Gene Ther. 2022. PMID: 35156839 Free PMC article.
-
Neutralizing antibodies against adeno-associated virus examined prospectively in pediatric patients with hemophilia.Gene Ther. 2012 Mar;19(3):288-94. doi: 10.1038/gt.2011.90. Epub 2011 Jun 23. Gene Ther. 2012. PMID: 21697954
-
Gene Delivery of Activated Factor VII Using Alternative Adeno-Associated Virus Serotype Improves Hemostasis in Hemophiliac Mice with FVIII Inhibitors and Adeno-Associated Virus Neutralizing Antibodies.Hum Gene Ther. 2017 Aug;28(8):654-666. doi: 10.1089/hum.2017.016. Epub 2017 May 5. Hum Gene Ther. 2017. PMID: 28478688 Free PMC article.
-
Clinical Considerations for Capsid Choice in the Development of Liver-Targeted AAV-Based Gene Transfer.Mol Ther Methods Clin Dev. 2019 Sep 10;15:170-178. doi: 10.1016/j.omtm.2019.08.015. eCollection 2019 Dec 13. Mol Ther Methods Clin Dev. 2019. PMID: 31660419 Free PMC article. Review.
-
Emerging Immunogenicity and Genotoxicity Considerations of Adeno-Associated Virus Vector Gene Therapy for Hemophilia.J Clin Med. 2021 Jun 2;10(11):2471. doi: 10.3390/jcm10112471. J Clin Med. 2021. PMID: 34199563 Free PMC article. Review.
Cited by
-
Monitoring cell-mediated immune responses in AAV gene therapy clinical trials using a validated IFN-γ ELISpot method.Mol Ther Methods Clin Dev. 2021 May 29;22:183-195. doi: 10.1016/j.omtm.2021.05.012. eCollection 2021 Sep 10. Mol Ther Methods Clin Dev. 2021. PMID: 34485604 Free PMC article.
-
Global Seroprevalence of Pre-existing Immunity Against AAV5 and Other AAV Serotypes in People with Hemophilia A.Hum Gene Ther. 2022 Apr;33(7-8):432-441. doi: 10.1089/hum.2021.287. Epub 2022 Mar 16. Hum Gene Ther. 2022. PMID: 35156839 Free PMC article.
-
Recombinant adeno-associated virus 8 vector in gene therapy: Opportunities and challenges.Genes Dis. 2023 Mar 24;11(1):283-293. doi: 10.1016/j.gendis.2023.02.010. eCollection 2024 Jan. Genes Dis. 2023. PMID: 37588223 Free PMC article. Review.
-
The seroprevalence of neutralizing antibodies against the adeno-associated virus capsids in Japanese hemophiliacs.Mol Ther Methods Clin Dev. 2022 Oct 27;27:404-414. doi: 10.1016/j.omtm.2022.10.014. eCollection 2022 Dec 8. Mol Ther Methods Clin Dev. 2022. PMID: 36381300 Free PMC article.
-
Screening data from 19 patients with late-onset Pompe disease for a phase I clinical trial of AAV8 vector-mediated gene therapy.JIMD Rep. 2023 Aug 17;64(5):393-400. doi: 10.1002/jmd2.12391. eCollection 2023 Sep. JIMD Rep. 2023. PMID: 37701327 Free PMC article.
References
-
- Petrini P. Identifying and overcoming barriers to prophylaxis in the management of hemophilia. Hemophilia. 2007;13(Suppl 2):16–22. - PubMed
-
- Farrugia A, Cassar J, Kimber MC, Bansal M, Fischer K, Auserswald G, et al. Treatment for life for severe hemophilia A‐ A cost‐utility model for prophylaxis vs. on‐demand treatment. Hemophilia. 2013;19:e228–38. - PubMed
-
- Chuah MK, Evens H, VandenDriessche T. Gene therapy for hemophilia. J Thromb Haemost. 2013;11:99–110. - PubMed
-
- Srivastava A, Brewer AK, Mauser‐Bunschoten EP, Key NS, Kitchen S, Llinas A, et al. Guidelines for the management of hemophilia. Hemophilia. 2013;19:e1–47. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical