A lumped parameter model of endoplasm flow in Physarum polycephalum explains migration and polarization-induced asymmetry during the onset of locomotion
- PMID: 31013306
- PMCID: PMC6478327
- DOI: 10.1371/journal.pone.0215622
A lumped parameter model of endoplasm flow in Physarum polycephalum explains migration and polarization-induced asymmetry during the onset of locomotion
Abstract
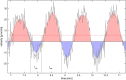
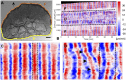
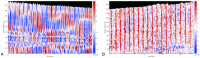
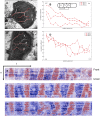
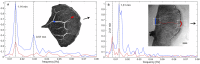
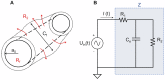
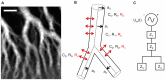
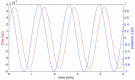
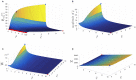
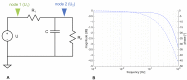
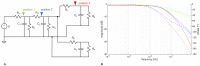
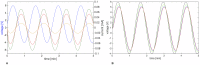
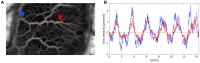
The plasmodial slime mold Physarum polycephalum exhibits strong, periodic flow of cytoplasm through the veins of its network. In the special case of mesoplasmodia, a newly described starvation-induced, shape-constant morphotype, this periodic endoplasm streaming is the basis of locomotion. Furthermore, we presume that cytoplasm flow is also involved in signal transmission and signal processing. Mesoplasmodia motility resembles amoeboid locomotion. In contrast to other amoebae, however, mesoplasmodia move without extending pseudopods and retain a coherent, fan-shaped morphology throughout their steady locomotion. Attaining sizes of up to 2 mm2, mesoplasmodia are also much bigger than other amoebae. We characterize this particular type of locomotion and identify patterns of movement. By using the analogy between pulsatile fluid flow through a network of elastic tubes and electrical circuits, we build a lumped model that explains observed fluid flow patterns. Essentially, the mesoplasmodium acts as a low-pass filter, permitting only low-frequency oscillations to propagate from back to front. This frequency selection serves to optimize flow and reduces power dissipation. Furthermore, we introduce a distributed element into the lumped model to explain cell polarization during the onset of chemotaxis: Biochemical cues (internal or external) lead to a local softening of the actin cortex, which in turn causes an increased flow of cytoplasm into that area and, thus, a net forward movement. We conclude that the internal actin-enclosed vein network gives the slime mold a high measure of control over fluid transport, especially by softening or hardening, which in turn leads to polarization and net movement.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


Similar articles
-
Different Morphotypes of Physarum polycephalum as Models for Chemotaxis and Locomotion.Methods Mol Biol. 2024;2828:69-78. doi: 10.1007/978-1-0716-4023-4_7. Methods Mol Biol. 2024. PMID: 39147971
-
Peristaltic transport and mixing of cytosol through the whole body of Physarum plasmodium.Math Med Biol. 2012 Sep;29(3):263-81. doi: 10.1093/imammb/dqr010. Epub 2011 Jul 12. Math Med Biol. 2012. PMID: 21750121
-
Random network peristalsis in Physarum polycephalum organizes fluid flows across an individual.Proc Natl Acad Sci U S A. 2013 Aug 13;110(33):13306-11. doi: 10.1073/pnas.1305049110. Epub 2013 Jul 29. Proc Natl Acad Sci U S A. 2013. PMID: 23898203 Free PMC article.
-
Fluid flows shaping organism morphology.Philos Trans R Soc Lond B Biol Sci. 2018 May 26;373(1747):20170112. doi: 10.1098/rstb.2017.0112. Philos Trans R Soc Lond B Biol Sci. 2018. PMID: 29632264 Free PMC article. Review.
-
Brainless but Multi-Headed: Decision Making by the Acellular Slime Mould Physarum polycephalum.J Mol Biol. 2015 Nov 20;427(23):3734-43. doi: 10.1016/j.jmb.2015.07.007. Epub 2015 Jul 17. J Mol Biol. 2015. PMID: 26189159 Review.
Cited by
-
Mitochondrial numbers increase during glucose deprivation in the slime mold Physarum polycephalum.Protoplasma. 2019 Nov;256(6):1647-1655. doi: 10.1007/s00709-019-01410-1. Epub 2019 Jul 2. Protoplasma. 2019. PMID: 31267225 Free PMC article.
-
Different Morphotypes of Physarum polycephalum as Models for Chemotaxis and Locomotion.Methods Mol Biol. 2024;2828:69-78. doi: 10.1007/978-1-0716-4023-4_7. Methods Mol Biol. 2024. PMID: 39147971
-
Adaptive behaviour and learning in slime moulds: the role of oscillations.Philos Trans R Soc Lond B Biol Sci. 2021 Mar 15;376(1820):20190757. doi: 10.1098/rstb.2019.0757. Epub 2021 Jan 25. Philos Trans R Soc Lond B Biol Sci. 2021. PMID: 33487112 Free PMC article.
-
Encoding memory in tube diameter hierarchy of living flow network.Proc Natl Acad Sci U S A. 2021 Mar 9;118(10):e2007815118. doi: 10.1073/pnas.2007815118. Proc Natl Acad Sci U S A. 2021. PMID: 33619174 Free PMC article.
-
Tracking Mechanical Stress and Cell Migration with Inexpensive Polymer Thin-Film Sensors.Adv Mater Interfaces. 2023 Jan 17;10(2):2201808. doi: 10.1002/admi.202201808. Epub 2022 Nov 18. Adv Mater Interfaces. 2023. PMID: 36817827 Free PMC article.
References
-
- Baranowski Z, Wohlfarth-Bottermann KE. Endoplasmic veins from plasmodia of Physarum polycephalum: a new strand model defined age, structure, and behavior. Eur J Cell Biol. 1982. April;27(1):1–9. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources

