Molecular Characterization of a Dirofilaria immitis Cysteine Protease Inhibitor (Cystatin) and Its Possible Role in Filarial Immune Evasion
- PMID: 31013806
- PMCID: PMC6523577
- DOI: 10.3390/genes10040300
Molecular Characterization of a Dirofilaria immitis Cysteine Protease Inhibitor (Cystatin) and Its Possible Role in Filarial Immune Evasion
Abstract
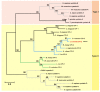
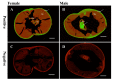
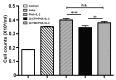
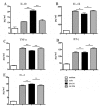
Infection with canine heartworm (Dirofilaria immitis), spread via mosquito vectors, causes coughing, asthma, pneumonia, and bronchitis in humans and other animals. The disease is especially severe and often fatal in dogs and represents a serious threat to public health worldwide. Cysteine protease inhibitors (CPIs), also known as cystatins, are major immunomodulators of the host immune response during nematode infections. Herein, we cloned and expressed the cystatin Di-CPI from D. immitis. Sequence analysis revealed two specific cystatin-like domains, a Q-x-V-x-G motif, and a SND motif. Phylogenetic analysis indicates that Di-CPI is a member of the second subgroup of nematode type II cystatins. Probing of D. immitis total proteins with anti-rDi-CPI polyclonal antibody revealed a weak signal, and immunofluorescence-based histochemical analysis showed that native Di-CPI is mainly localized in the cuticle of male and female worms and the gut of male worms. Treatment of canine peripheral blood mononuclear cells (PMBCs) with recombinant Di-CPI induced a Th2-type immune response characterized by high expression of the anti-inflammatory factor interleukin-10. Proliferation assays showed that Di-CPI inhibits the proliferation of canine PMBCs by 15%. Together, the results indicate that Di-CPI might be related to cellular hyporesponsiveness in dirofilariasis and may help D. immitis to evade the host immune system.
Keywords: Dirofilaria immitis; cystatin; immune evasion; immunohistochemistry; peripheral blood mononuclear cells.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Cloning and preliminary characterization of a novel cuticular antigen from the filarial parasite Dirofilaria immitis.Parasitol Int. 2000 Dec;49(4):321-5. doi: 10.1016/s1383-5769(00)00055-6. Parasitol Int. 2000. PMID: 11077266
-
Dirofilaria immitis encodes Di-nhr-7, a putative orthologue of the Drosophila ecdysone-regulated E78 gene.Mol Biochem Parasitol. 2002 Feb;119(2):169-77. doi: 10.1016/s0166-6851(01)00412-1. Mol Biochem Parasitol. 2002. PMID: 11814569
-
Antibody to the Dirofilaria immitis aspartyl protease inhibitor homologue is a diagnostic marker for feline heartworm infections.J Parasitol. 1998 Dec;84(6):1231-6. J Parasitol. 1998. PMID: 9920319
-
Cystatins from filarial parasites: evolution, adaptation and function in the host-parasite relationship.Int J Biochem Cell Biol. 2008;40(6-7):1389-98. doi: 10.1016/j.biocel.2007.11.012. Epub 2007 Dec 4. Int J Biochem Cell Biol. 2008. PMID: 18249028 Review.
-
Immunomodulatory properties of cystatins.Cell Mol Life Sci. 2002 Sep;59(9):1503-12. doi: 10.1007/s00018-002-8525-4. Cell Mol Life Sci. 2002. PMID: 12440772 Free PMC article. Review.
Cited by
-
Evolutionary Analysis of Cystatins of Early-Emerging Metazoans Reveals a Novel Subtype in Parasitic Cnidarians.Biology (Basel). 2021 Feb 3;10(2):110. doi: 10.3390/biology10020110. Biology (Basel). 2021. PMID: 33546310 Free PMC article.
-
Cystatin from the helminth Ascaris lumbricoides upregulates mevalonate and cholesterol biosynthesis pathways and immunomodulatory genes in human monocyte-derived dendritic cells.Front Immunol. 2024 Feb 28;15:1328401. doi: 10.3389/fimmu.2024.1328401. eCollection 2024. Front Immunol. 2024. PMID: 38481989 Free PMC article.
References
-
- Dillon R.A. Influence of number of parasites and exercise on the severity of heartworm disease in dogs. Proc. Heartworm Symp. 1995;113:95.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

