Modelling the long-term benefits of tolvaptan therapy on renal function decline in autosomal dominant polycystic kidney disease: an exploratory analysis using the ADPKD outcomes model
- PMID: 31014270
- PMCID: PMC6480528
- DOI: 10.1186/s12882-019-1290-5
Modelling the long-term benefits of tolvaptan therapy on renal function decline in autosomal dominant polycystic kidney disease: an exploratory analysis using the ADPKD outcomes model
Abstract
Background: The short-term efficacy of tolvaptan in patients with autosomal dominant polycystic kidney disease (ADPKD) has been demonstrated across several phase 3 trials, while the ADPKD Outcomes Model (ADPKD-OM) represents a validated approach to predict natural disease progression over a lifetime horizon. This study describes the implementation of a tolvaptan treatment effect within the ADPKD-OM and explores the potential long-term benefits of tolvaptan therapy in ADPKD.
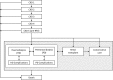
Methods: The effect of tolvaptan on ADPKD progression was modelled by applying a constant treatment effect to the rate of renal function decline, consistent with that observed in the Tolvaptan Efficacy and Safety in Management of Autosomal Dominant Polycystic Kidney Disease and Its Outcomes trial (TEMPO 3:4; ClinicalTrials.gov identifier NCT00428948 ). Predictions generated by the ADPKD-OM were compared against aggregated data from a subsequent extension trial (TEMPO 4:4; ClinicalTrials.gov identifier NCT01214421 ) and the Replicating Evidence of Preserved Renal Function an Investigation of Tolvaptan Safety Efficacy in ADPKD trial (REPRISE; ClinicalTrials.gov identifier NCT02160145 ). Following validation, an application of the ADPKD-OM sought to estimate the benefit of tolvaptan therapy on time to end-stage renal disease (ESRD), in a range of ADPKD populations.
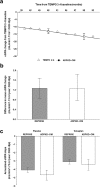
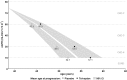
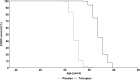
Results: Model validation against TEMPO 4:4 and REPRISE demonstrated the accuracy and generalisability of the tolvaptan treatment effect applied within the ADPKD-OM. In simulated patients matched to the overall TEMPO 3:4 trial population at baseline, tolvaptan therapy was predicted to delay the mean age of ESRD onset by five years, compared to natural disease progression (57 years versus 52 years, respectively). In subgroup and sensitivity analyses, the estimated delay to ESRD was greatest among patients with CKD stage 1 at baseline (6.6 years), compared to CKD 2 and 3 subgroups (4.7 and 2.7 years, respectively); and ADPKD patients in Mayo subclasses 1C-1E.
Conclusions: This study demonstrated the potential for tolvaptan therapy to delay time to ESRD, particularly among patients with early-stage CKD and evidence of rapidly progressing disease. Data arising from this study highlight the value to be gained by early intervention and long-term treatment with tolvaptan, which may alleviate the economic and societal costs of providing care to patients who progress to ESRD.
Keywords: Autosomal dominant polycystic kidney disease; Disease modelling; End-stage renal disease; Renal function decline; Tolvaptan.
Conflict of interest statement
Ethics approval and consent to participate
This article is based on previously conducted research and does not involve any new studies of human or animal subjects performed by any of the authors, as such ethics approval and consent to participate were not required.
Consent for publication
Not applicable.
Competing interests
HBW, PM and KH have served as consultants to and received research funding from Otsuka Pharmaceutical Europe Ltd. in relation to this study. KO is an employee of Otsuka Pharmaceutical Europe Ltd.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures

References
-
- Spithoven EM, Kramer A, Meijer E, Orskov B, Wanner C, Abad JM, et al. Renal replacement therapy for autosomal dominant polycystic kidney disease (ADPKD) in Europe: prevalence and survival—an analysis of data from the ERA-EDTA registry. Nephrol Dial Transplant. 2014;29(suppl 4):iv15–iv25. doi: 10.1093/ndt/gfu017. - DOI - PMC - PubMed
-
- Gansevoort RT, Arici M, Benzing T, Birn H, Capasso G, Covic A, et al. Recommendations for the use of tolvaptan in autosomal dominant polycystic kidney disease: a position statement on behalf of the ERA-EDTA working groups on inherited kidney disorders and European renal best practice. Nephrol Dial Transplant. 2016;31(3):337–348. doi: 10.1093/ndt/gfv456. - DOI - PMC - PubMed
-
- Pharmaceuticals and medical devices agency. Review report: Samsca 2014 [28 Feburary 2018]. Available from: http://www.pmda.go.jp/files/000208511.pdf
-
- European Medicines Agency. European public assessment report - product information: Jinarc (tolvaptan) 2017 [16 January 2018]. Available from: http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Info....
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

