Mechanisms of action of ruxolitinib in murine models of hemophagocytic lymphohistiocytosis
- PMID: 31015190
- PMCID: PMC6624972
- DOI: 10.1182/blood.2019000761
Mechanisms of action of ruxolitinib in murine models of hemophagocytic lymphohistiocytosis
Abstract
Hemophagocytic lymphohistiocytosis (HLH) is an often-fatal disorder characterized by the overactivation of T cells and macrophages that excessively produce proinflammatory cytokines, including interferon-γ (IFN-γ). Previously, we reported that the JAK inhibitor ruxolitinib dampens T-cell activation and lessens inflammation in a model of HLH in which perforin-deficient (Prf1 -/-) mice are infected with lymphocytic choriomeningitis virus (LCMV). Ruxolitinib inhibits signaling downstream of IFN-γ, as well as several other JAK-dependent cytokines. As a consequence, it remained unclear whether ruxolitinib was exerting its beneficial effects in HLH by inhibiting IFN-γ signaling or by targeting signaling initiated by other proinflammatory cytokines. To address this question, we compared the effects of ruxolitinib with those obtained using an IFN-γ-neutralizing antibody (αIFN-γ) in 2 murine HLH models. In both models, ruxolitinib and αIFN-γ reduced inflammation-associated anemia, indicating that ruxolitinib operates in an IFN-γ-dependent manner to reverse this HLH manifestation. In contrast, the number and activation status of T cells and neutrophils, as well as their infiltration into tissues, were significantly reduced following treatment with ruxolitinib, but they remained unchanged or were increased following treatment with αIFN-γ. Notably, despite discontinuation of ruxolitinib, LCMV-infected Prf1 -/- mice exhibited enhanced survival compared with mice in which αIFN-γ was discontinued. This protective effect could be mimicked by transient treatment with αIFN-γ and a neutrophil-depleting antibody. Thus, ruxolitinib operates through IFN-γ-dependent and -independent mechanisms to dampen HLH by targeting the deleterious effects of T cells and neutrophils, with the latter representing an unappreciated and understudied cell type that contributes to HLH pathogenesis.
© 2019 by The American Society of Hematology.
Conflict of interest statement
Conflict-of-interest disclosure: K.E.N. receives research funding from Incyte Pharmaceuticals and Alpine Biosciences. The remaining authors declare no competing financial interests
Figures


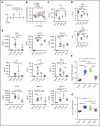

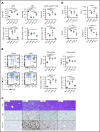
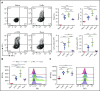

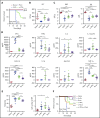
Comment in
-
Calming the storm in HLH.Blood. 2019 Jul 11;134(2):103-104. doi: 10.1182/blood.2019001333. Blood. 2019. PMID: 31296541 No abstract available.
References
-
- Janka GE, Lehmberg K. Hemophagocytic syndromes--an update. Blood Rev. 2014;28(4):135-142. - PubMed
-
- Akashi K, Hayashi S, Gondo H, et al. . Involvement of interferon-gamma and macrophage colony-stimulating factor in pathogenesis of haemophagocytic lymphohistiocytosis in adults. Br J Haematol. 1994;87(2):243-250. - PubMed
-
- Osugi Y, Hara J, Tagawa S, et al. . Cytokine production regulating Th1 and Th2 cytokines in hemophagocytic lymphohistiocytosis. Blood. 1997;89(11):4100-4103. - PubMed
-
- Yang SL, Xu XJ, Tang YM, et al. . Associations between inflammatory cytokines and organ damage in pediatric patients with hemophagocytic lymphohistiocytosis. Cytokine. 2016;85:14-17. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

