TORC1 modulation in adipose tissue is required for organismal adaptation to hypoxia in Drosophila
- PMID: 31015407
- PMCID: PMC6478872
- DOI: 10.1038/s41467-019-09643-7
TORC1 modulation in adipose tissue is required for organismal adaptation to hypoxia in Drosophila
Abstract
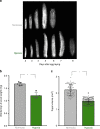
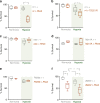
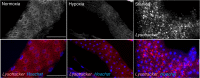
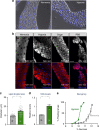
Animals often develop in environments where conditions such as food, oxygen and temperature fluctuate. The ability to adapt their metabolism to these fluctuations is important for normal development and viability. In most animals, low oxygen (hypoxia) is deleterious. However some animals can alter their physiology to tolerate hypoxia. Here we show that TORC1 modulation in adipose tissue is required for organismal adaptation to hypoxia in Drosophila. We find that hypoxia rapidly suppresses TORC1 signaling in Drosophila larvae via TSC-mediated inhibition of Rheb. We show that this hypoxia-mediated inhibition of TORC1 specifically in the larval fat body is essential for viability. Moreover, we find that these effects of TORC1 inhibition on hypoxia tolerance are mediated through remodeling of fat body lipid storage. These studies identify the larval adipose tissue as a key hypoxia-sensing tissue that coordinates whole-body development and survival to changes in environmental oxygen by modulating TORC1 and lipid metabolism.
Conflict of interest statement
The authors declare no competing interests.
Figures




Similar articles
-
FoxO restricts growth and differentiation of cells with elevated TORC1 activity under nutrient restriction.PLoS Genet. 2018 Apr 20;14(4):e1007347. doi: 10.1371/journal.pgen.1007347. eCollection 2018 Apr. PLoS Genet. 2018. PMID: 29677182 Free PMC article.
-
The TSC1/2 complex controls Drosophila pigmentation through TORC1-dependent regulation of catecholamine biosynthesis.PLoS One. 2012;7(11):e48720. doi: 10.1371/journal.pone.0048720. Epub 2012 Nov 7. PLoS One. 2012. PMID: 23144943 Free PMC article.
-
Sima, a Drosophila homolog of HIF-1α, in fat body tissue inhibits larval body growth by inducing Tribbles gene expression.Genes Cells. 2022 Feb;27(2):145-151. doi: 10.1111/gtc.12913. Epub 2021 Dec 27. Genes Cells. 2022. PMID: 34918430
-
As the fat flies: The dynamic lipid droplets of Drosophila embryos.Biochim Biophys Acta. 2015 Sep;1851(9):1156-85. doi: 10.1016/j.bbalip.2015.04.002. Epub 2015 Apr 13. Biochim Biophys Acta. 2015. PMID: 25882628 Free PMC article. Review.
-
Cellular and developmental adaptations to hypoxia: a Drosophila perspective.Methods Enzymol. 2007;435:123-44. doi: 10.1016/S0076-6879(07)35007-6. Methods Enzymol. 2007. PMID: 17998052 Review.
Cited by
-
Early-life hypoxia alters adult physiology and reduces stress resistance and lifespan in Drosophila.J Exp Biol. 2020 Nov 23;223(Pt 22):jeb226027. doi: 10.1242/jeb.226027. J Exp Biol. 2020. PMID: 32988998 Free PMC article.
-
Compartment and cell-type specific hypoxia responses in the developing Drosophila brain.Biol Open. 2020 Aug 18;9(8):bio053629. doi: 10.1242/bio.053629. Biol Open. 2020. PMID: 32816692 Free PMC article.
-
Tolerance to Hypoxia Is Promoted by FOXO Regulation of the Innate Immunity Transcription Factor NF-κB/Relish in Drosophila.Genetics. 2020 Aug;215(4):1013-1025. doi: 10.1534/genetics.120.303219. Epub 2020 Jun 8. Genetics. 2020. PMID: 32513813 Free PMC article.
-
Transcriptome analysis of FOXO-dependent hypoxia gene expression identifies Hipk as a regulator of low oxygen tolerance in Drosophila.G3 (Bethesda). 2022 Dec 1;12(12):jkac263. doi: 10.1093/g3journal/jkac263. G3 (Bethesda). 2022. PMID: 36200850 Free PMC article.
-
What can the common fruit fly teach us about stroke?: lessons learned from the hypoxic tolerant Drosophila melanogaster.Front Cell Neurosci. 2024 Mar 22;18:1347980. doi: 10.3389/fncel.2024.1347980. eCollection 2024. Front Cell Neurosci. 2024. PMID: 38584778 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

