Correlative 3D Imaging and Microfluidic Modelling of Human Pulmonary Lymphatics using Immunohistochemistry and High-resolution μCT
- PMID: 31015547
- PMCID: PMC6478691
- DOI: 10.1038/s41598-019-42794-7
Correlative 3D Imaging and Microfluidic Modelling of Human Pulmonary Lymphatics using Immunohistochemistry and High-resolution μCT
Abstract
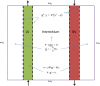
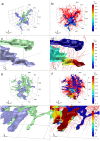
Lung lymphatics maintain fluid homoeostasis by providing a drainage system that returns fluid, cells and metabolites to the circulatory system. The 3D structure of the human pulmonary lymphatic network is essential to lung function, but it is poorly characterised. Image-based 3D mathematical modelling of pulmonary lymphatic microfluidics has been limited by the lack of accurate and representative image geometries. This is due to the microstructural similarity of the lymphatics to the blood vessel network, the lack of lymphatic-specific biomarkers, the technical limitations associated with image resolution in 3D, and sectioning artefacts present in 2D techniques. We present a method that combines lymphatic specific (D240 antibody) immunohistochemistry (IHC), optimised high-resolution X-ray microfocus computed tomography (μCT) and finite-element mathematical modelling to assess the function of human peripheral lung tissue. The initial results identify lymphatic heterogeneity within and between lung tissue. Lymphatic vessel volume fraction and fractal dimension significantly decreases away from the lung pleural surface (p < 0.001, n = 25 and p < 0.01, n = 20, respectively). Microfluidic modelling successfully shows that in lung tissue the fluid derived from the blood vessels drains through the interstitium into the lymphatic vessel network and this drainage is different in the subpleural space compared to the intralobular space. When comparing lung tissue from health and disease, human pulmonary lymphatics were significantly different across five morphometric measures used in this study (p ≤ 0.0001). This proof of principle study establishes a new engineering technology and workflow for further studies of pulmonary lymphatics and demonstrates for the first time the combination of correlative μCT and IHC to enable 3D mathematical modelling of human lung microfluidics at micrometre resolution.
Conflict of interest statement
The authors declare no competing interests.
Figures




References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

