Synergetic Functional Nanocomposites Enhance Immunotherapy in Solid Tumors by Remodeling the Immunoenvironment
- PMID: 31016114
- PMCID: PMC6469336
- DOI: 10.1002/advs.201802012
Synergetic Functional Nanocomposites Enhance Immunotherapy in Solid Tumors by Remodeling the Immunoenvironment
Abstract
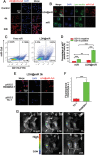
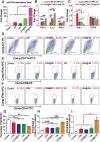
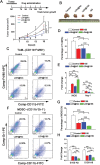
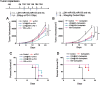
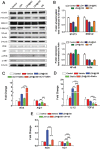
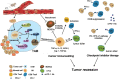
Checkpoint blockade immunotherapy has demonstrated significant clinical success in various malignant tumors. However, the therapeutic response is limited due to the immunosuppressive tumor microenvironment (ITM). In this study, a functional nanomaterial, layered double hydroxides (LDHs), carrying specific functional miR155 is developed to modulate ITM by synergistically repolarizing tumor associated macrophages (TAMs) to M1 subtype. LDH nanoparticles loaded with miR155 (LDH@155) exhibit superior ability in cellular uptake by murine macrophages, miR escape into the cytoplasm and TAMs specific delivery when introtumoral administration. Meanwhile, upon exposure to LDH@155, TAMs are significantly skewed to M1 subtype, which markedly inhibits myeloid-derived suppressor cells (MDSCs) formation and stimulates T-lymphocytes to secrete more interferon-γ (IFN-γ) cytokines in vitro. Introtumoral administration of LDH@155 reduces the percentage of TAMs and MDSCs in the tumor and elevates CD4+ and CD8+ T cell infiltration and activation, which can promote therapeutic efficiency of α-PD-1 antibody immunotherapy. Furthermore, it is found that LDH@155 significantly decreases the expression level of phosphorylated STAT3 and ERK1/2 and activates NF-κB expression in TAMs, indicating that the STAT3, ERK1/2, and NF-κB signaling pathways may involve in LDH@155-induced macrophage polarization. Overall, the results suggest that LDH@155 nanoparticles may, in the future, function as a promising agent for cancer combinational immunotherapy.
Keywords: immunosuppressive tumor microenvironments; immunotherapy; layered double hydroxides; microRNA; nanoparticles.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
