Correlating Infant Fecal Microbiota Composition and Human Milk Oligosaccharide Consumption by Microbiota of 1-Month-Old Breastfed Infants
- PMID: 31017343
- PMCID: PMC6618098
- DOI: 10.1002/mnfr.201801214
Correlating Infant Fecal Microbiota Composition and Human Milk Oligosaccharide Consumption by Microbiota of 1-Month-Old Breastfed Infants
Abstract
Scope: Understanding the biological functions of human milk oligosaccharides (HMOs) in shaping gastrointestinal (GI) tract microbiota during infancy is of great interest. A link between HMOs in maternal milk and infant fecal microbiota composition is examined and the role of microbiota in degrading HMOs within the GI tract of healthy, breastfed, 1-month-old infants is investigated.
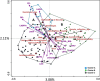
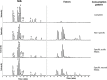
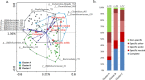
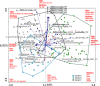
Methods and results: Maternal breast milk and infant feces are from the KOALA Birth Cohort. HMOs are quantified in milk and infant fecal samples using liquid chromatography-mass spectrometry. Fecal microbiota composition is characterized using Illumina HiSeq 16S rRNA gene amplicon sequencing. The composition is associated with gender, delivery mode, and milk HMOs: Lacto-N-fucopentaose I and 2'-fucosyllactose. Overall, Bifidobacterium, Bacteroides, Escherichia-Shigella, and Parabacteroides are predominating genera. Three different patterns in infant fecal microbiota structure are detected. GI degradation of HMOs is strongly associated with fecal microbiota composition, and there is a link between utilization of specific HMOs and relative abundance of various phylotypes (operational taxonomic units).
Conclusions: HMOs in maternal milk are among the important factors shaping GI tract microbiota in 1-month-old breastfed infants. An infant's ability to metabolize different HMOs strongly correlates with fecal microbiota composition and specifically with phylotypes within genera Bifidobacterium, Bacteroides, and Lactobacillus.
Keywords: breastfeeding; human milk oligosaccharide; microbial clusters; microbiome.
© 2019 The Authors. Published by WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Linking Human Milk Oligosaccharides, Infant Fecal Community Types, and Later Risk To Require Antibiotics.mBio. 2020 Mar 17;11(2):e03196-19. doi: 10.1128/mBio.03196-19. mBio. 2020. PMID: 32184252 Free PMC article. Clinical Trial.
-
Maternal Fucosyltransferase 2 Status Associates with the Profiles of Human Milk Oligosaccharides and the Fecal Microbiota Composition of Breastfed Infants.J Agric Food Chem. 2021 Mar 17;69(10):3032-3043. doi: 10.1021/acs.jafc.0c04575. Epub 2021 Mar 6. J Agric Food Chem. 2021. PMID: 33677972
-
Fecal microbiota composition of breast-fed infants is correlated with human milk oligosaccharides consumed.J Pediatr Gastroenterol Nutr. 2015 Jun;60(6):825-33. doi: 10.1097/MPG.0000000000000752. J Pediatr Gastroenterol Nutr. 2015. PMID: 25651488 Free PMC article.
-
Factors affecting early-life intestinal microbiota development.Nutrition. 2020 Oct;78:110812. doi: 10.1016/j.nut.2020.110812. Epub 2020 Mar 25. Nutrition. 2020. PMID: 32464473 Review.
-
Varied Pathways of Infant Gut-Associated Bifidobacterium to Assimilate Human Milk Oligosaccharides: Prevalence of the Gene Set and Its Correlation with Bifidobacteria-Rich Microbiota Formation.Nutrients. 2019 Dec 26;12(1):71. doi: 10.3390/nu12010071. Nutrients. 2019. PMID: 31888048 Free PMC article. Review.
Cited by
-
Human milk oligosaccharides, antimicrobial drugs, and the gut microbiota of term neonates: observations from the KOALA birth cohort study.Gut Microbes. 2023 Jan-Dec;15(1):2164152. doi: 10.1080/19490976.2022.2164152. Gut Microbes. 2023. PMID: 36617628 Free PMC article.
-
Interactions between human milk oligosaccharides, microbiota and immune factors in milk of women with and without mastitis.Sci Rep. 2022 Jan 25;12(1):1367. doi: 10.1038/s41598-022-05250-7. Sci Rep. 2022. PMID: 35079053 Free PMC article.
-
Building Robust Assemblages of Bacteria in the Human Gut in Early Life.Appl Environ Microbiol. 2021 Oct 28;87(22):e0144921. doi: 10.1128/AEM.01449-21. Epub 2021 Sep 1. Appl Environ Microbiol. 2021. PMID: 34469198 Free PMC article. Review.
-
The Infant Gut Commensal Bacteroides dorei Presents a Generalized Transcriptional Response to Various Human Milk Oligosaccharides.Front Cell Infect Microbiol. 2022 Mar 18;12:854122. doi: 10.3389/fcimb.2022.854122. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 35372092 Free PMC article.
-
Effect of breast milk with or without bacteria on infant gut microbiota.BMC Pregnancy Childbirth. 2022 Jul 26;22(1):595. doi: 10.1186/s12884-022-04930-6. BMC Pregnancy Childbirth. 2022. PMID: 35883060 Free PMC article.
References
-
- Rautava S., Luoto R., Salminen S., Isolauri E., Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 565. - PubMed
-
- Zivkovic A. M., Lewis Z. T., German J. B., Mills D. A., Funct. Food Rev. 2013, 5, 3.
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

