Drosophila Temperature Preference Rhythms: An Innovative Model to Understand Body Temperature Rhythms
- PMID: 31018551
- PMCID: PMC6514862
- DOI: 10.3390/ijms20081988
Drosophila Temperature Preference Rhythms: An Innovative Model to Understand Body Temperature Rhythms
Abstract
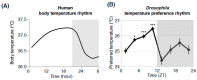
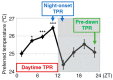
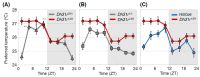
Human body temperature increases during wakefulness and decreases during sleep. The body temperature rhythm (BTR) is a robust output of the circadian clock and is fundamental for maintaining homeostasis, such as generating metabolic energy and sleep, as well as entraining peripheral clocks in mammals. However, the mechanisms that regulate BTR are largely unknown. Drosophila are ectotherms, and their body temperatures are close to ambient temperature; therefore, flies select a preferred environmental temperature to set their body temperature. We identified a novel circadian output, the temperature preference rhythm (TPR), in which the preferred temperature in flies increases during the day and decreases at night. TPR, thereby, produces a daily BTR. We found that fly TPR shares many features with mammalian BTR. We demonstrated that diuretic hormone 31 receptor (DH31R) mediates Drosophila TPR and that the closest mouse homolog of DH31R, calcitonin receptor (Calcr), is essential for mice BTR. Importantly, both TPR and BTR are regulated in a distinct manner from locomotor activity rhythms, and neither DH31R nor Calcr regulates locomotor activity rhythms. Our findings suggest that DH31R/Calcr is an ancient and specific mediator of BTR. Thus, understanding fly TPR will provide fundamental insights into the molecular and neural mechanisms that control BTR in mammals.
Keywords: Calcitonin receptor; Calcr; DH31; DH31R; Drosophila; PDF; PDFR; body temperature rhythms; circadian rhythms; locomotor activity rhythms.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Molecular and Neural Mechanisms of Temperature Preference Rhythm in Drosophila melanogaster.J Biol Rhythms. 2023 Aug;38(4):326-340. doi: 10.1177/07487304231171624. Epub 2023 May 24. J Biol Rhythms. 2023. PMID: 37222551 Free PMC article. Review.
-
Calcitonin receptors are ancient modulators for rhythms of preferential temperature in insects and body temperature in mammals.Genes Dev. 2018 Jan 15;32(2):140-155. doi: 10.1101/gad.307884.117. Epub 2018 Feb 12. Genes Dev. 2018. PMID: 29440246 Free PMC article.
-
Drosophila DH31 Neuropeptide and PDF Receptor Regulate Night-Onset Temperature Preference.J Neurosci. 2016 Nov 16;36(46):11739-11754. doi: 10.1523/JNEUROSCI.0964-16.2016. J Neurosci. 2016. PMID: 27852781 Free PMC article.
-
Design and analysis of temperature preference behavior and its circadian rhythm in Drosophila.J Vis Exp. 2014 Jan 13;(83):e51097. doi: 10.3791/51097. J Vis Exp. 2014. PMID: 24457268 Free PMC article.
-
Circadian Rhythms and Sleep in Drosophila melanogaster.Genetics. 2017 Apr;205(4):1373-1397. doi: 10.1534/genetics.115.185157. Genetics. 2017. PMID: 28360128 Free PMC article. Review.
Cited by
-
Dynamic encoding of temperature in the central circadian circuit coordinates physiological activities.Nat Commun. 2024 Apr 2;15(1):2834. doi: 10.1038/s41467-024-47278-5. Nat Commun. 2024. PMID: 38565846 Free PMC article.
-
Conformational flexibility of tubulin dimers regulates the transitions of microtubule dynamic instability.bioRxiv [Preprint]. 2025 Jul 2:2025.06.30.662375. doi: 10.1101/2025.06.30.662375. bioRxiv. 2025. PMID: 40631249 Free PMC article. Preprint.
-
Taste triggers a homeostatic temperature control in hungry flies.Elife. 2024 Dec 2;13:RP94703. doi: 10.7554/eLife.94703. Elife. 2024. PMID: 39621014 Free PMC article.
-
Time-Restricted G-Protein Signaling Pathways via GPR176, Gz, and RGS16 Set the Pace of the Master Circadian Clock in the Suprachiasmatic Nucleus.Int J Mol Sci. 2020 Jul 17;21(14):5055. doi: 10.3390/ijms21145055. Int J Mol Sci. 2020. PMID: 32709014 Free PMC article. Review.
-
Dorsal clock networks drive temperature preference rhythms in Drosophila.Cell Rep. 2022 Apr 12;39(2):110668. doi: 10.1016/j.celrep.2022.110668. Cell Rep. 2022. PMID: 35417715 Free PMC article.
References
-
- Aschoff J. Circadian Control of Body-Temperature. J. Therm. Biol. 1983;8:143–147. doi: 10.1016/0306-4565(83)90094-3. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

