SR-B1 drives endothelial cell LDL transcytosis via DOCK4 to promote atherosclerosis
- PMID: 31019307
- PMCID: PMC6631346
- DOI: 10.1038/s41586-019-1140-4
SR-B1 drives endothelial cell LDL transcytosis via DOCK4 to promote atherosclerosis
Abstract
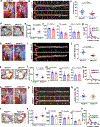
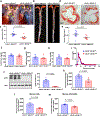
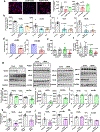
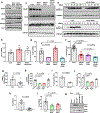
Atherosclerosis, which underlies life-threatening cardiovascular disorders such as myocardial infarction and stroke1, is initiated by passage of low-density lipoprotein (LDL) cholesterol into the artery wall and its engulfment by macrophages, which leads to foam cell formation and lesion development2,3. It is unclear how circulating LDL enters the artery wall to instigate atherosclerosis. Here we show in mice that scavenger receptor class B type 1 (SR-B1) in endothelial cells mediates the delivery of LDL into arteries and its accumulation by artery wall macrophages, thereby promoting atherosclerosis. LDL particles are colocalized with SR-B1 in endothelial cell intracellular vesicles in vivo, and transcytosis of LDL across endothelial monolayers requires its direct binding to SR-B1 and an eight-amino-acid cytoplasmic domain of the receptor that recruits the guanine nucleotide exchange factor dedicator of cytokinesis 4 (DOCK4)4. DOCK4 promotes internalization of SR-B1 and transport of LDL by coupling the binding of LDL to SR-B1 with activation of RAC1. The expression of SR-B1 and DOCK4 is increased in atherosclerosis-prone regions of the mouse aorta before lesion formation, and in human atherosclerotic arteries when compared with normal arteries. These findings challenge the long-held concept that atherogenesis involves passive movement of LDL across a compromised endothelial barrier. Interventions that inhibit the endothelial delivery of LDL into artery walls may represent a new therapeutic category in the battle against cardiovascular disease.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures









Comment in
-
Active LDL trafficking drives atherosclerosis.Nat Rev Cardiol. 2019 Jul;16(7):384. doi: 10.1038/s41569-019-0210-5. Nat Rev Cardiol. 2019. PMID: 31068687 No abstract available.
References
-
- Tabas I, Williams KJ, & Boren J Subendothelial lipoprotein retention as the initiating process in atherosclerosis: update and therapeutic implications. Circulation 116, 1832–1844 (2007). - PubMed
-
- Gadea G & Blangy A Dock-family exchange factors in cell migration and disease. Eur. J. Cell Biol 93, 466–477 (2014). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

