The Response to DNA Damage at Telomeric Repeats and Its Consequences for Telomere Function
- PMID: 31022960
- PMCID: PMC6523756
- DOI: 10.3390/genes10040318
The Response to DNA Damage at Telomeric Repeats and Its Consequences for Telomere Function
Abstract
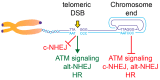
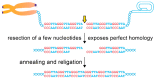
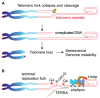
Telomeric repeats, coated by the shelterin complex, prevent inappropriate activation of the DNA damage response at the ends of linear chromosomes. Shelterin has evolved distinct solutions to protect telomeres from different aspects of the DNA damage response. These solutions include formation of t-loops, which can sequester the chromosome terminus from DNA-end sensors and inhibition of key steps in the DNA damage response. While blocking the DNA damage response at chromosome ends, telomeres make wide use of many of its players to deal with exogenous damage and replication stress. This review focuses on the interplay between the end-protection functions and the response to DNA damage occurring inside the telomeric repeats, as well as on the consequences that telomere damage has on telomere structure and function.
Keywords: alternative lengthening of telomeres; end-protection problem; shelterin complex; telomere damage; telomere maintenance; telomere replication; telomeric double strand breaks.
Conflict of interest statement
I have no competing financial interests.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

