Constitutive expression and cell-surface display of a bacterial β-mannanase in Lactobacillus plantarum
- PMID: 31023309
- PMCID: PMC6482533
- DOI: 10.1186/s12934-019-1124-y
Constitutive expression and cell-surface display of a bacterial β-mannanase in Lactobacillus plantarum
Abstract
Background: Lactic acid bacteria (LAB) are important microorganisms in the food and beverage industry. Due to their food-grade status and probiotic characteristics, several LAB are considered as safe and effective cell-factories for food-application purposes. In this present study, we aimed at constitutive expression of a mannanase from Bacillus licheniformis DSM13, which was subsequently displayed on the cell surface of Lactobacillus plantarum WCFS1, for use as whole-cell biocatalyst in oligosaccharide production.
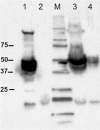
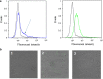
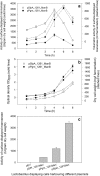
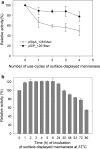
Results: Two strong constitutive promoters, Pgm and SlpA, from L. acidophilus NCFM and L. acidophilus ATCC4356, respectively, were used to replace the inducible promoter in the lactobacillal pSIP expression system for the construction of constitutive pSIP vectors. The mannanase-encoding gene (manB) was fused to the N-terminal lipoprotein anchor (Lp_1261) from L. plantarum and the resulting fusion protein was cloned into constitutive pSIP vectors and expressed in L. plantarum WCFS1. The localization of the protein on the bacterial cell surface was confirmed by flow cytometry and immunofluorescence microscopy. The mannanase activity and the reusability of the constructed L. plantarum displaying cells were evaluated. The highest mannanase activities on the surface of L. plantarum cells obtained under the control of the Pgm and SlpA promoters were 1200 and 3500 U/g dry cell weight, respectively, which were 2.6- and 7.8-fold higher compared to the activity obtained from inducible pSIP anchoring vectors. Surface-displayed mannanase was shown to be able to degrade galactomannan into manno-oligosaccharides (MOS).
Conclusion: This work demonstrated successful displaying of ManB on the cell surface of L. plantarum WCFS1 using constitutive promoter-based anchoring vectors for use in the production of manno-oligosaccharides, which are potentially prebiotic compounds with health-promoting effects. Our approach, where the enzyme of interest is displayed on the cell surface of a food-grade organism with the use of strong constitutive promoters, which continuously drive synthesis of the recombinant protein without the need to add an inducer or change the growth conditions of the host strain, should result in the availability of safe, stable food-grade biocatalysts.
Keywords: Cell-surface display; Constitutive promoter; Lactobacillus plantarum; Lipoprotein anchor; Mannanase; Pgm; SlpA; Whole-cell biocatalyst.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


References
-
- Khanongnuch C, Sa-nguansook C, Lumyong S. Nutritive quality of mannanase treated copra meal in broiler diets and effectiveness on some fecal bacteria. Int J Poult Sci. 2006;5:1087–1091. doi: 10.3923/ijps.2006.1087.1091. - DOI
-
- Blibech M, Chaari F, Bhiri F, Dammak I, Ghorbel RE, Chaabouni SE. Production of manno-oligosaccharides from locust bean gum using immobilized Penicillium occitanis mannanase. J Mol Catal B Enzym. 2011;73:111–115.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

