Re-expression of SynGAP protein in adulthood improves translatable measures of brain function and behavior
- PMID: 31025938
- PMCID: PMC6504227
- DOI: 10.7554/eLife.46752
Re-expression of SynGAP protein in adulthood improves translatable measures of brain function and behavior
Abstract
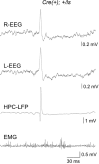
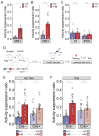
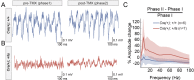
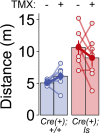
It remains unclear to what extent neurodevelopmental disorder (NDD) risk genes retain functions into adulthood and how they may influence disease phenotypes. SYNGAP1 haploinsufficiency causes a severe NDD defined by autistic traits, cognitive impairment, and epilepsy. To determine if this gene retains therapeutically-relevant biological functions into adulthood, we performed a gene restoration technique in a mouse model for SYNGAP1 haploinsufficiency. Adult restoration of SynGAP protein improved behavioral and electrophysiological measures of memory and seizure. This included the elimination of interictal events that worsened during sleep. These events may be a biomarker for generalized cortical dysfunction in SYNGAP1 disorders because they also worsened during sleep in the human patient population. We conclude that SynGAP protein retains biological functions throughout adulthood and that non-developmental functions may contribute to disease phenotypes. Thus, treatments that target debilitating aspects of severe NDDs, such as medically-refractory seizures and cognitive impairment, may be effective in adult patients.
Keywords: autism; biomarker; circuits; epilepsy; human; mouse; mouse model; neuroscience; reversal.
© 2019, Creson et al.
Conflict of interest statement
TC, CR, EH, TV, MK, AJ, JH, JT, CM, GR No competing interests declared, LC Reviewing editor, eLife
Figures





References
-
- Aceti M, Creson TK, Vaissiere T, Rojas C, Huang WC, Wang YX, Petralia RS, Page DT, Miller CA, Rumbaugh G. Syngap1 haploinsufficiency damages a postnatal critical period of pyramidal cell structural maturation linked to cortical circuit assembly. Biological Psychiatry. 2015;77:805–815. doi: 10.1016/j.biopsych.2014.08.001. - DOI - PMC - PubMed
-
- Berryer MH, Hamdan FF, Klitten LL, Møller RS, Carmant L, Schwartzentruber J, Patry L, Dobrzeniecka S, Rochefort D, Neugnot-Cerioli M, Lacaille JC, Niu Z, Eng CM, Yang Y, Palardy S, Belhumeur C, Rouleau GA, Tommerup N, Immken L, Beauchamp MH, Patel GS, Majewski J, Tarnopolsky MA, Scheffzek K, Hjalgrim H, Michaud JL, Di Cristo G. Mutations in SYNGAP1 cause intellectual disability, autism, and a specific form of epilepsy by inducing haploinsufficiency. Human Mutation. 2013;34:385–394. doi: 10.1002/humu.22248. - DOI - PubMed
-
- Carvill GL, Heavin SB, Yendle SC, McMahon JM, O'Roak BJ, Cook J, Khan A, Dorschner MO, Weaver M, Calvert S, Malone S, Wallace G, Stanley T, Bye AM, Bleasel A, Howell KB, Kivity S, Mackay MT, Rodriguez-Casero V, Webster R, Korczyn A, Afawi Z, Zelnick N, Lerman-Sagie T, Lev D, Møller RS, Gill D, Andrade DM, Freeman JL, Sadleir LG, Shendure J, Berkovic SF, Scheffer IE, Mefford HC. Targeted resequencing in epileptic encephalopathies identifies de novo mutations in CHD2 and SYNGAP1. Nature Genetics. 2013;45:825–830. doi: 10.1038/ng.2646. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 NS064079/NS/NINDS NIH HHS/United States
- MH108408/MH/NIMH NIH HHS/United States
- DA036376/DA/NIDA NIH HHS/United States
- MH102450/MH/NIMH NIH HHS/United States
- T32 MH106454/MH/NIMH NIH HHS/United States
- NS064079/NS/NINDS NIH HHS/United States
- MH105400/MH/NIMH NIH HHS/United States
- K08 NS091381/NS/NINDS NIH HHS/United States
- MH096847/MH/NIMH NIH HHS/United States
- R01 MH108408/MH/NIMH NIH HHS/United States
- Mentored Clinical Scientist Research Career Development Award NS091381/NS/NINDS NIH HHS/United States
- T32DA01892/DA/NIDA NIH HHS/United States
- DA034116/DA/NIDA NIH HHS/United States
- R01 MH102450/MH/NIMH NIH HHS/United States
- R21 DA036376/DA/NIDA NIH HHS/United States
- R01 MH096847/MH/NIMH NIH HHS/United States
- R01 DA034116/DA/NIDA NIH HHS/United States
- NS100738/NS/NINDS NIH HHS/United States
- R01 MH105400/MH/NIMH NIH HHS/United States
- R56 NS100738/NS/NINDS NIH HHS/United States
- R01 NS100738/NS/NINDS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

