Metal-Catalyzed Photooxidation of Flavones in Aqueous Media
- PMID: 31031566
- PMCID: PMC6474262
- DOI: 10.1002/ejic.201800288
Metal-Catalyzed Photooxidation of Flavones in Aqueous Media
Abstract
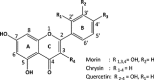
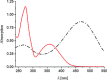
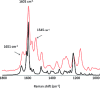
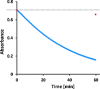
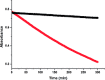
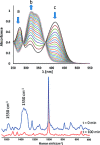
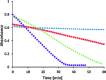
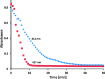
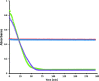
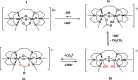
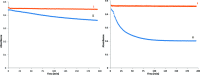
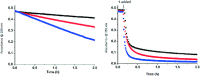
Soluble model compounds, such as flavones, are frequently employed in initial and mechanistic studies under homogeneous conditions in the search for effective bleaching catalysts for raw cotton. The relevance of model substrates, such as morin and chrysin, and especially their reactivity with manganese catalysts [i.e. in combination with 1,4,7-triazacyclononane (tacn) based ligands] applied in raw cotton bleaching with H2O2 in alkaline solutions is examined. We show that morin, used frequently as a model, is highly sensitive to oxidation with O2, by processes catalyzed by trace metal ions, that can be accelerated photochemically, although not involve generation of 1O2. The structurally related chrysin is not susceptible to such photo-accelerated oxidation with O2. Furthermore, chrysin is oxidized by H2O2 only in the presence of a Mn-tacn based catalyst, and does not undergo oxidation with O2 as terminal oxidant. Chrysin mimics the behavior of raw cotton's chromophores in their catalyzed oxidation with H2O2, and is likely a mechanistically relevant model compound for the study of transition metal catalysts for dye bleaching catalysts under homogeneous conditions.
Keywords: Bleaching; Chrysin; Manganese; Morin; Oxidation; Photochemistry.
Figures
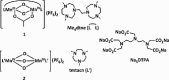
Similar articles
-
Bioinspired Manganese and Iron Complexes for Enantioselective Oxidation Reactions: Ligand Design, Catalytic Activity, and Beyond.Acc Chem Res. 2019 Aug 20;52(8):2370-2381. doi: 10.1021/acs.accounts.9b00285. Epub 2019 Jul 23. Acc Chem Res. 2019. PMID: 31333021
-
Synthetic applications of nonmetal catalysts for homogeneous oxidations.Chem Rev. 2001 Nov;101(11):3499-548. doi: 10.1021/cr000019k. Chem Rev. 2001. PMID: 11840992
-
Comparative study of the catalytic activity of [Mn(II)(bpy)2Cl2] and [Mn2(III/IV)(mu-O)2(bpy)4](ClO4)3 in the H2O2 induced oxidation of organic dyes in carbonate buffered aqueous solution.Dalton Trans. 2010 Apr 7;39(13):3264-72. doi: 10.1039/b925160j. Epub 2010 Feb 11. Dalton Trans. 2010. PMID: 20449456
-
Review of iron-free Fenton-like systems for activating H2O2 in advanced oxidation processes.J Hazard Mater. 2014 Jun 30;275:121-35. doi: 10.1016/j.jhazmat.2014.04.054. Epub 2014 May 2. J Hazard Mater. 2014. PMID: 24857896 Review.
-
Activation of Dioxygen by Iron and Manganese Complexes: A Heme and Nonheme Perspective.J Am Chem Soc. 2016 Sep 14;138(36):11410-28. doi: 10.1021/jacs.6b05251. Epub 2016 Aug 30. J Am Chem Soc. 2016. PMID: 27576170 Free PMC article. Review.
Cited by
-
Biocompatible Hyaluronic Acid-Stabilized Copper Nanoparticles for the Selective Oxidation of Morin Dye by H2O2.ACS Omega. 2025 Apr 2;10(14):14431-14438. doi: 10.1021/acsomega.5c00769. eCollection 2025 Apr 15. ACS Omega. 2025. PMID: 40256537 Free PMC article.
References
-
- Weissermal H. and Arpe K., in: Industrial Organic Chemistry, VCH‐Wiley, Weinheim, 1997.
-
- Hage R. and Lienke A., Angew. Chem. Int. Ed., 2005, 45, 206–222; - PubMed
- Angew. Chem., 2005, 117, 212–229.
-
- Beychok M. R., Atmos. Environ., 1987, 21, 29–36.
-
- Nakamata K. and Ohi H., J. Wood Sci., 2003, 49, 525–530.
-
- Nakamata K., Motoe Y. and Ohi H., J. Wood Sci., 2004, 50, 242–247.
LinkOut - more resources
Full Text Sources
