Copper Ligand and Anion Effects: Controlling the Kinetics of the Photoinitiated Copper(I) Catalyzed Azide-Alkyne Cycloaddition Polymerization
- PMID: 31031838
- PMCID: PMC6483394
- DOI: 10.1039/C8PY01004H
Copper Ligand and Anion Effects: Controlling the Kinetics of the Photoinitiated Copper(I) Catalyzed Azide-Alkyne Cycloaddition Polymerization
Abstract
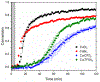
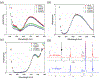
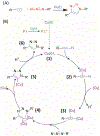
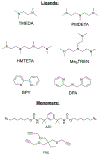
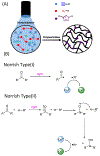
The kinetics of photoinduced copper(I) catalyzed azide-alkyne cycloaddition (CuAAC) polymerizations were assessed as a function of copper(II) amine-based ligands. Copper(II) bromide ligated with 1,1,4,7,10,10-hexamethylenetetramine (HMTETA) exhibited the fastest kinetics in both Norrish type(I) and type(II) photoinitiating systems. A characteristic induction period is observed with these polymerizations and is manipulated by adding an external tertiary amine in Norrish Type(II) photoinitating systems or by changing the anion of the copper(II) salt. Halides, specifically bromide and chloride, exhibit the fastest kinetics with the smallest induction period in comparison with organic anions, such as bistriflimide and triflate. The temporal control of the photo-CuAAC polymerization is affected by pre-ligation of the copper catalyst, by the presence of certain anions such as acetate, and by specific ligands such as tetramethylethylenediamine (TMEDA).
Conflict of interest statement
Conflicts of interest There are no conflicts to declare.
Figures


Similar articles
-
Kinetics of bulk photo-initiated copper(i)-catalyzed azide-alkyne cycloaddition (CuAAC) polymerizations.Polym Chem. 2016 Jan 21;7(3):603-612. doi: 10.1039/c5py01655j. Epub 2015 Nov 18. Polym Chem. 2016. PMID: 27429650 Free PMC article.
-
Reduced shrinkage stress via photo-initiated copper(I)-catalyzed cycloaddition polymerizations of azide-alkyne resins.Dent Mater. 2016 Nov;32(11):1332-1342. doi: 10.1016/j.dental.2016.07.014. Epub 2016 Aug 11. Dent Mater. 2016. PMID: 27524230 Free PMC article.
-
Photoinitiated Copper(I) Catalyzed Azide-Alkyne Cycloaddition Reaction for Ion Conductive Networks.ACS Macro Lett. 2019;8(7):795-799. doi: 10.1021/acsmacrolett.9b00324. Epub 2019 Jun 17. ACS Macro Lett. 2019. PMID: 32864190 Free PMC article.
-
Advancements in the mechanistic understanding of the copper-catalyzed azide-alkyne cycloaddition.Beilstein J Org Chem. 2013 Dec 2;9:2715-50. doi: 10.3762/bjoc.9.308. Beilstein J Org Chem. 2013. PMID: 24367437 Free PMC article. Review.
-
In-situ Generated and Premade 1-Copper(I) Alkynes in Cycloadditions.Chem Rec. 2017 Dec;17(12):1231-1248. doi: 10.1002/tcr.201700011. Epub 2017 Jun 22. Chem Rec. 2017. PMID: 28639363 Review.
Cited by
-
Effects of ring-strain on the ultrafast photochemistry of cyclic ketones.Chem Sci. 2020 Jan 13;11(7):1991-2000. doi: 10.1039/c9sc05208a. Chem Sci. 2020. PMID: 34123294 Free PMC article.
-
CuAAC-methacrylate interpenetrating polymer network (IPN) properties modulated by visible-light photoinitiation.Polym Chem. 2023 Aug 28;14(32):3739-3748. doi: 10.1039/d3py00507k. Epub 2023 Jul 28. Polym Chem. 2023. PMID: 37663952 Free PMC article.
-
Photoclick Chemistry: A Bright Idea.Chem Rev. 2021 Jun 23;121(12):6915-6990. doi: 10.1021/acs.chemrev.0c01212. Epub 2021 Apr 9. Chem Rev. 2021. PMID: 33835796 Free PMC article. Review.
References
-
- Rostovtsev VV, Green LG, Fokin VV and Sharpless KB, Angew. Chemie - Int. Ed, 2002, 41, 2596–2599. - PubMed
-
- Tornøe CW, Christensen C and Meldal M, J. Org. Chem, 2002, 67, 3057–3064. - PubMed
-
- Kolb HC, Finn MG and Sharpless KB, Angew. Chemie - Int. Ed, 2001, 40, 2004–2021. - PubMed
-
- Kolb HC and Sharpless KB, Drug Discov. Today, 2003, 8, 1128–1137. - PubMed
-
- Tsatsakis AM, Nawaz MA, Kouretas D, Balias G, Savolainen K, Tutelyan VA, Golokhvast KS, Lee JD, Yang SH and Chung G, Environ. Res, 2017, 156, 818–833. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
