Whole body glucoregulation and tissue-specific glucose uptake in a novel Akt substrate of 160 kDa knockout rat model
- PMID: 31034517
- PMCID: PMC6488193
- DOI: 10.1371/journal.pone.0216236
Whole body glucoregulation and tissue-specific glucose uptake in a novel Akt substrate of 160 kDa knockout rat model
Abstract
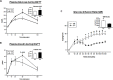
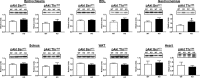
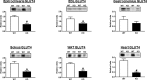
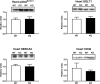
Akt substrate of 160 kDa (also called AS160 or TBC1D4) is a Rab GTPase activating protein and key regulator of insulin-stimulated glucose uptake which is expressed by multiple tissues, including skeletal muscle, white adipose tissue (WAT) and the heart. This study introduces a novel rat AS160-knockout (AS160-KO) model that was created using CRISPR/Cas9 technology. We compared male AS160-KO versus wildtype (WT) rats for numerous metabolism-related endpoints. Body mass, body composition, energy expenditure and physical activity did not differ between genotypes. Oral glucose intolerance was detected in AS160-KO versus WT rats (P<0.005). A hyperinsulinemic-euglycemic clamp (HEC) revealed insulin resistance for glucose infusion rate (P<0.05) with unaltered hepatic glucose production in AS160-KO versus WT rats. Genotype-effects on glucose uptake during the HEC: 1) was significantly lower in epitrochlearis (P<0.01) and extensor digitorum longus (P<0.05) of AS160-KO versus WT rats, and tended to be lower for AS160-KO versus WT rats in the soleus (P<0.06) and gastrocnemius (P<0.08); 2) tended to be greater for AS160-KO versus WT rats in white adipose tissue (P = 0.09); and 3) was significantly greater in the heart (P<0.005) of AS160-KO versus WT rats. GLUT4 protein abundance was significantly lower for AS160-KO versus WT rats in each tissue analyzed (P<0.01-0.001) except the gastrocnemius. Ex vivo insulin-stimulated glucose uptake was significantly lower (P<0.001) for AS160-KO versus WT rats in isolated epitrochlearis or soleus. Insulin-stimulated Akt phosphorylation (in vivo or ex vivo) did not differ between genotypes for any tissue tested. Ex vivo AICAR-stimulated glucose uptake by isolated epitrochlearis was significantly lower for AS160-KO versus WT rats (P<0.01) without genotype-induced alteration in AMP-activated protein phosphorylation. This unique AS160-KO rat model, which elucidated striking genotype-related modifications in glucoregulation, will enable future research aimed at understanding AS160's roles in numerous physiological processes in response to various interventions (e.g., diet and/or exercise).
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures






Similar articles
-
In vivo glucoregulation and tissue-specific glucose uptake in female Akt substrate 160 kDa knockout rats.PLoS One. 2020 Feb 13;15(2):e0223340. doi: 10.1371/journal.pone.0223340. eCollection 2020. PLoS One. 2020. PMID: 32053588 Free PMC article.
-
AS160 expression, but not AS160 Serine-588, Threonine-642, and Serine-704 phosphorylation, is essential for elevated insulin-stimulated glucose uptake by skeletal muscle from female rats after acute exercise.FASEB J. 2023 Jul;37(7):e23021. doi: 10.1096/fj.202300282RR. FASEB J. 2023. PMID: 37289137 Free PMC article.
-
Prior treatment with the AMPK activator AICAR induces subsequently enhanced glucose uptake in isolated skeletal muscles from 24-month-old rats.Appl Physiol Nutr Metab. 2018 Aug;43(8):795-805. doi: 10.1139/apnm-2017-0858. Epub 2018 Mar 8. Appl Physiol Nutr Metab. 2018. PMID: 29518344 Free PMC article.
-
Role of Akt substrate of 160 kDa in insulin-stimulated and contraction-stimulated glucose transport.Appl Physiol Nutr Metab. 2007 Jun;32(3):557-66. doi: 10.1139/H07-026. Appl Physiol Nutr Metab. 2007. PMID: 17510697 Review.
-
Roles of TBC1D1 and TBC1D4 in insulin- and exercise-stimulated glucose transport of skeletal muscle.Diabetologia. 2015 Jan;58(1):19-30. doi: 10.1007/s00125-014-3395-5. Epub 2014 Oct 4. Diabetologia. 2015. PMID: 25280670 Free PMC article. Review.
Cited by
-
Novel Bacillus ginsengihumi CMRO6 Inhibits Adipogenesis via p38MAPK/Erk44/42 and Stimulates Glucose Uptake in 3T3-L1 Pre-Adipocytes through Akt/AS160 Signaling.Int J Mol Sci. 2022 Apr 25;23(9):4727. doi: 10.3390/ijms23094727. Int J Mol Sci. 2022. PMID: 35563118 Free PMC article.
-
Excess Accumulation of Lipid Impairs Insulin Sensitivity in Skeletal Muscle.Int J Mol Sci. 2020 Mar 12;21(6):1949. doi: 10.3390/ijms21061949. Int J Mol Sci. 2020. PMID: 32178449 Free PMC article. Review.
-
Complex rearrangement in TBC1D4 in an individual with diabetes due to severe insulin resistance syndrome.Eur J Hum Genet. 2024 Feb;32(2):232-237. doi: 10.1038/s41431-023-01512-8. Epub 2023 Dec 12. Eur J Hum Genet. 2024. PMID: 38086948 Free PMC article.
-
Fiber type-specific effects of acute exercise on insulin-stimulated AS160 phosphorylation in insulin-resistant rat skeletal muscle.Am J Physiol Endocrinol Metab. 2019 Dec 1;317(6):E984-E998. doi: 10.1152/ajpendo.00304.2019. Epub 2019 Oct 1. Am J Physiol Endocrinol Metab. 2019. PMID: 31573845 Free PMC article.
-
Exercise effects on γ3-AMPK activity, phosphorylation of Akt2 and AS160, and insulin-stimulated glucose uptake in insulin-resistant rat skeletal muscle.J Appl Physiol (1985). 2020 Feb 1;128(2):410-421. doi: 10.1152/japplphysiol.00428.2019. Epub 2020 Jan 16. J Appl Physiol (1985). 2020. PMID: 31944891 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

