Influence of SHH/GLI1 axis on EMT mediated migration and invasion of breast cancer cells
- PMID: 31036836
- PMCID: PMC6488587
- DOI: 10.1038/s41598-019-43093-x
Influence of SHH/GLI1 axis on EMT mediated migration and invasion of breast cancer cells
Abstract
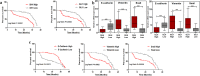
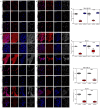
Sonic Hedgehog signaling is critical for breast morphogenesis and cancer. The present study was conducted to explore the influence of SHH/GLI1 axis on epithelial mesenchymal transition and invasion in breast cancer cells. SHH/GLI1 positive samples demonstrated high expression of Snail and Vimentin with relatively low expression of E-cadherin. Overexpression of Vimentin and Snail in SHH/GLI1 positive patients was also associated with poor overall survival. Interestingly, GANT61 (GLI1 inhibitor) exposure significantly reduced cell viability and induced apoptosis at 10 µM. Suppression of Hedgehog pathway either by CRISPR mediated SHH knock out or GANT61 altered regulation of EMT markers in breast cancer cells. Moreover, in-activation of SHH/GLI1 axis also significantly restricted cell migration and invasiveness. These findings suggest that targeting SHH/GLI1 axis alters expression of EMT markers and abrogates neoplastic invasion in breast cancer cells.
Conflict of interest statement
The authors declare no competing interests.
Figures





References
-
- Sasaki H, Nishizaki Y, Hui C, Nakafuku M, Kondoh H. Regulation of Gli2 and Gli3 activities by an amino-terminal repression domain: implication of Gli2 and Gli3 as primary mediators of Shh signaling. Development. 1999;126:3915–24. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

