5α-cyprinol sulfate, a bile salt from fish, induces diel vertical migration in Daphnia
- PMID: 31045492
- PMCID: PMC6559785
- DOI: 10.7554/eLife.44791
5α-cyprinol sulfate, a bile salt from fish, induces diel vertical migration in Daphnia
Abstract
Prey are under selection to minimize predation losses. In aquatic environments, many prey use chemical cues released by predators, which initiate predator avoidance. A prominent example of behavioral predator-avoidance constitutes diel vertical migration (DVM) in the freshwater microcrustacean Daphnia spp., which is induced by chemical cues (kairomones) released by planktivorous fish. In a bioassay-guided approach using liquid chromatography and mass spectrometry, we identified the kairomone from fish incubation water as 5α-cyprinol sulfate inducing DVM in Daphnia at picomolar concentrations. The role of 5α-cyprinol sulfate in lipid digestion in fish explains why from an evolutionary perspective fish has not stopped releasing 5α-cyprinol sulfate despite the disadvantages for the releaser. The identification of the DVM-inducing kairomone enables investigating its spatial and temporal distribution and the underlying molecular mechanism of its perception. Furthermore, it allows to test if fish-mediated inducible defenses in other aquatic invertebrates are triggered by the same compound.
Keywords: Cyprinus carpio; Daphnia magna; Rutilus rutilus; daphnia; diel vertical migration; ecology; kairomone; zooplankton.
© 2019, Hahn et al.
Conflict of interest statement
MH, CE, LB, Ev No competing interests declared
Figures
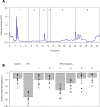
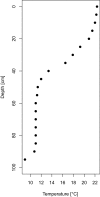



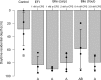






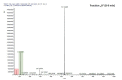

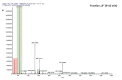


Comment in
-
Finding the fish factor.Elife. 2019 Jun 11;8:e48459. doi: 10.7554/eLife.48459. Elife. 2019. PMID: 31184305 Free PMC article.
References
-
- Abrams PA. Foraging time optimization and interactions in food webs. The American Naturalist. 1984;124:80–96. doi: 10.1086/284253. - DOI
-
- Berendonk TU. Influence of fish kairomones on the ovipositing behavior of Chaoborus imagines. Limnology and Oceanography. 1999;44:454–458. doi: 10.4319/lo.1999.44.2.0454. - DOI
-
- Bjærke O, Andersen T, Titelman J. Predator chemical cues increase growth and alter development in nauplii of a marine copepod. Marine Ecology Progress Series. 2014;510:15–24. doi: 10.3354/meps10918. - DOI
-
- Bollens SM, Frost BW. Diel vertical migration in zooplankton: rapid individual response to predators. Journal of Plankton Research. 1991;13:1359–1365. doi: 10.1093/plankt/13.6.1359. - DOI
-
- Brönmark C, Hansson L-A. Chemical Ecology in Aquatic Systems. Oxford University Press; 2012.
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

