Early-life stress alters sleep structure and the excitatory-inhibitory balance in the nucleus accumbens in aged mice
- PMID: 31045908
- PMCID: PMC6616240
- DOI: 10.1097/CM9.0000000000000279
Early-life stress alters sleep structure and the excitatory-inhibitory balance in the nucleus accumbens in aged mice
Abstract
Background: Exposure to adverse experiences in early life may profoundly reshape the neurodevelopmental trajectories of the brain and lead to long-lasting behavioral and neural alterations. One deleterious effect of early-life stress that manifests in later life is sleep disturbance, but this has not been examined in aged mice and the underlying neural mechanisms remain unknown. Considering the important role of the nucleus accumbens (NAc) in the sleep-wake regulation, this study aimed to assess the effects of early-life stress on the sleep behaviors in aged mice and the potential involvement of the NAc in stress-induced sleep abnormalities.
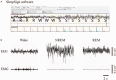
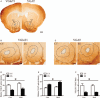
Methods: Twenty aged male C57BL/6 mice (>16 months, n = 10 per group) were used in this study. During post-natal days 2 to 9, dams were provided with either sufficient (control) or a limited nesting and bedding materials (stressed). When the mice were 16 to 17 months old, their sleep-wake behaviors were recorded over 24 h using electroencephalogram and electromyelogram. The amount of each sleep-wake stage, mean duration, and stage transition was analyzed. Then, five animals were randomly chosen from each group and were used to measure the expression levels of vesicular glutamate transporter-1 (VGluT1) and vesicular transporters of γ-aminobutyric acid (VGAT) in the NAc using immunohistochemistry. Group comparisons were carried out using Student t test or analysis of variances when appropriate.
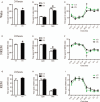
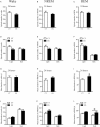
Results: Compared with the control mice, the early-life stressed aged mice spent less time awake over 24 h (697.97 ± 77.47 min vs. 631.33 ± 34.73 min, t17 = 2.376, P = 0.030), accordingly, non-rapid eye movement sleep time was increased (667.37 ± 62.07 min vs. 723.54 ± 39.21 min, t17 = 2.326, P = 0.033) and mean duration of rapid eye movement sleep was prolonged (73.00 ± 8.98 min vs. 89.39 ± 12.69 min, t17 = 3.277, P = 0.004). Meanwhile, we observed decreased VGluT1/VGAT ratios in the NAc in the stressed group (F(1, 16) = 81.04, P < 0.001).
Conclusion: Early adverse experiences disrupt sleep behaviors in aged mice, which might be associated with the excitatory-inhibitory imbalance in the NAc.
Figures
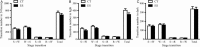
Similar articles
-
The Nucleus Accumbens CRH-CRHR1 System Mediates Early-Life Stress-Induced Sleep Disturbance and Dendritic Atrophy in the Adult Mouse.Neurosci Bull. 2023 Jan;39(1):41-56. doi: 10.1007/s12264-022-00903-z. Epub 2022 Jun 25. Neurosci Bull. 2023. PMID: 35750984 Free PMC article.
-
Adolescent stress increases depression-like behaviors and alters the excitatory-inhibitory balance in aged mice.Chin Med J (Engl). 2019 Jul 20;132(14):1689-1699. doi: 10.1097/CM9.0000000000000313. Chin Med J (Engl). 2019. PMID: 31268909 Free PMC article.
-
Increased vulnerability to depressive-like behavior of mice with decreased expression of VGLUT1.Biol Psychiatry. 2009 Aug 1;66(3):275-82. doi: 10.1016/j.biopsych.2009.02.027. Epub 2009 May 1. Biol Psychiatry. 2009. PMID: 19409534
-
The control of sleep and wakefulness by mesolimbic dopamine systems.Neurosci Res. 2017 May;118:66-73. doi: 10.1016/j.neures.2017.04.008. Epub 2017 Apr 20. Neurosci Res. 2017. PMID: 28434991 Review.
-
[Selective stimulations and lesions of the rat brain nuclei as the models for research of the human sleep pathology mechanisms].Glas Srp Akad Nauka Med. 2011;(51):85-97. Glas Srp Akad Nauka Med. 2011. PMID: 22165729 Review. Serbian.
Cited by
-
Effects of Electroacupuncture on Sleep via the Dopamine System of the HPA Axis in Rats after Cage Change.Evid Based Complement Alternat Med. 2021 Jul 1;2021:5527060. doi: 10.1155/2021/5527060. eCollection 2021. Evid Based Complement Alternat Med. 2021. PMID: 34306138 Free PMC article.
-
Prenatal alcohol exposure alters mRNA expression for stress peptides, glucocorticoid receptor function and immune factors in acutely stressed neonatal brain.Front Neurosci. 2023 Jun 23;17:1203557. doi: 10.3389/fnins.2023.1203557. eCollection 2023. Front Neurosci. 2023. PMID: 37425005 Free PMC article.
-
The Arousal-motor Hypothesis of Dopamine Function: Evidence that Dopamine Facilitates Reward Seeking in Part by Maintaining Arousal.Neuroscience. 2022 Sep 1;499:64-103. doi: 10.1016/j.neuroscience.2022.07.008. Epub 2022 Jul 16. Neuroscience. 2022. PMID: 35853563 Free PMC article.
-
The Nucleus Accumbens CRH-CRHR1 System Mediates Early-Life Stress-Induced Sleep Disturbance and Dendritic Atrophy in the Adult Mouse.Neurosci Bull. 2023 Jan;39(1):41-56. doi: 10.1007/s12264-022-00903-z. Epub 2022 Jun 25. Neurosci Bull. 2023. PMID: 35750984 Free PMC article.
-
Transcutaneous Electrical Acupoint Stimulation Improves Postoperative Sleep Quality in Patients Undergoing Laparoscopic Gastrointestinal Tumor Surgery: A Prospective, Randomized Controlled Trial.Pain Ther. 2023 Jun;12(3):707-722. doi: 10.1007/s40122-023-00493-2. Epub 2023 Mar 14. Pain Ther. 2023. PMID: 36928500 Free PMC article.
References
-
- Palagini L, Drake CL, Gehrman P, Meerlo P, Riemann D. Early-life origin of adult insomnia: does prenatal-early-life stress play a role? Sleep Med 2015; 16:446–456. doi: 10.1016/j.sleep.2014.10.013. - PubMed
-
- Chapman DP, Wheaton AG, Anda RF, Croft JB, Edwards VJ, Liu Y, et al. Adverse childhood experiences and sleep disturbances in adults. Sleep Med 2011; 12:773–779. doi: 10.1016/j.sleep.2011.03.013. - PubMed
-
- Koskenvuo K, Hublin C, Partinen M, Paunio T, Koskenvuo M. Childhood adversities and quality of sleep in adulthood: a population-based study of 26,000 Finns. Sleep Med 2010; 11:17–22. doi: 10.1016/j.sleep.2009.03.010. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

