Straight to the heart: Pleiotropic antiarrhythmic actions of oral anticoagulants
- PMID: 31054953
- PMCID: PMC6987959
- DOI: 10.1016/j.phrs.2019.104257
Straight to the heart: Pleiotropic antiarrhythmic actions of oral anticoagulants
Abstract
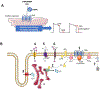
Mechanistic understanding of atrial fibrillation (AF) pathophysiology and the complex bidirectional relationship with thromboembolic risk remains limited. Oral anticoagulation is a mainstay of AF management. An emerging concept is that anticoagulants may themselves have potential pleiotropic disease-modifying effects. We here review the available evidence for hemostasis-independent actions of the oral anticoagulants on electrical and structural remodeling, and the inflammatory component of the vulnerable substrate.
Keywords: Anticoagulants; Atrial fibrillation; Calcium; Protease-activated receptors; Stroke; Thrombin.
Copyright © 2019 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Figures


Similar articles
-
Pleiotropic effects of factor Xa and thrombin: what to expect from novel anticoagulants.Cardiovasc Res. 2014 Mar 1;101(3):344-51. doi: 10.1093/cvr/cvt343. Epub 2014 Jan 2. Cardiovasc Res. 2014. PMID: 24385341 Review.
-
New drugs for atrial fibrillation.J Interv Card Electrophysiol. 2008 Oct;23(1):15-21. doi: 10.1007/s10840-008-9278-2. Epub 2008 Jun 6. J Interv Card Electrophysiol. 2008. PMID: 18535890 Review.
-
New and emerging antiarrhythmic and anticoagulant agents for atrial fibrillation.Am J Health Syst Pharm. 2010 May 1;67(9 Suppl 5):S26-34. doi: 10.2146/ajhp100154. Am J Health Syst Pharm. 2010. PMID: 20410533
-
Atrial fibrillation and human immunodeficiency virus type-1 infection: a systematic review. Implications for anticoagulant and antiarrhythmic therapy.Br J Clin Pharmacol. 2019 Mar;85(3):508-515. doi: 10.1111/bcp.13837. Epub 2019 Jan 11. Br J Clin Pharmacol. 2019. PMID: 30575989 Free PMC article.
-
Stroke prevention in atrial fibrillation: impact of novel oral anticoagulants.Clin Appl Thromb Hemost. 2013 Jun;19(3):241-8. doi: 10.1177/1076029612458148. Epub 2012 Sep 4. Clin Appl Thromb Hemost. 2013. PMID: 22952215 Review.
Cited by
-
Interaction of Cardiovascular Nonmodifiable Risk Factors, Comorbidities and Comedications With Ischemia/Reperfusion Injury and Cardioprotection by Pharmacological Treatments and Ischemic Conditioning.Pharmacol Rev. 2023 Jan;75(1):159-216. doi: 10.1124/pharmrev.121.000348. Epub 2022 Dec 8. Pharmacol Rev. 2023. PMID: 36753049 Free PMC article. Review.
-
Breaking bad: Higher risk of osteoporosis with vitamin K antagonists compared to direct oral anticoagulants in patients with atrial fibrillation.Int J Cardiol Heart Vasc. 2020 Apr 10;27:100487. doi: 10.1016/j.ijcha.2020.100487. eCollection 2020 Apr. Int J Cardiol Heart Vasc. 2020. PMID: 32310243 Free PMC article. No abstract available.
-
Natural history of COVID-19 and current knowledge on treatment therapeutic options.Biomed Pharmacother. 2020 Sep;129:110493. doi: 10.1016/j.biopha.2020.110493. Epub 2020 Jul 3. Biomed Pharmacother. 2020. PMID: 32768971 Free PMC article. Review.
-
Anticoagulant and antiarrhythmic effects of heparin in the treatment of COVID-19 patients.J Thromb Haemost. 2020 Aug;18(8):2073-2075. doi: 10.1111/jth.14902. J Thromb Haemost. 2020. PMID: 32408391 Free PMC article. No abstract available.
-
Thrombin receptor PAR4 cross-activates the tyrosine kinase c-met in atrial cardiomyocytes.Naunyn Schmiedebergs Arch Pharmacol. 2025 Mar;398(3):2783-2796. doi: 10.1007/s00210-024-03436-6. Epub 2024 Sep 16. Naunyn Schmiedebergs Arch Pharmacol. 2025. PMID: 39283528 Free PMC article.
References
-
- Kirchhof P, Breithardt G, Bax J, Benninger G, Blomstrom-Lundqvist C, Boriani G, Brandes A, Brown H, Brueckmann M, Calkins H, Calvert M, Christoffels V, Crijns H, Dobrev D, Ellinor P, Fabritz L, Fetsch T, Freedman SB, Gerth A, Goette A, Guasch E, Hack G, Haegeli L, Hatem S, Haeusler KG, Heidbuchel H, Heinrich-Nols J, Hidden-Lucet F, Hindricks G, Juul-Moller S, Kaab S, Kappenberger L, Kespohl S, Kotecha D, Lane DA, Leute A, Lewalter T, Meyer R, Mont L, Munzel F, Nabauer M, Nielsen JC, Oeff M, Oldgren J, Oto A, Piccini JP, Pilmeyer A, Potpara T, Ravens U, Reinecke H, Rostock T, Rustige J, Savelieva I, Schnabel R, Schotten U, Schwichtenberg L, Sinner MF, Steinbeck G, Stoll M, Tavazzi L, Themistoclakis S, Tse HF, Van Gelder IC, Vardas PE, Varpula T, Vincent A, Werring D, Willems S, Ziegler A, Lip GY Camm AJ, A roadmap to improve the quality of atrial fibrillation management: proceedings from the fifth Atrial Fibrillation Network/European Heart Rhythm Association consensus conference, Europace, 2016;18:37–50, 10.1093/europace/euv304 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

