Evolution of Cell Wall Polymers in Tip-Growing Land Plant Gametophytes: Composition, Distribution, Functional Aspects and Their Remodeling
- PMID: 31057570
- PMCID: PMC6482432
- DOI: 10.3389/fpls.2019.00441
Evolution of Cell Wall Polymers in Tip-Growing Land Plant Gametophytes: Composition, Distribution, Functional Aspects and Their Remodeling
Abstract
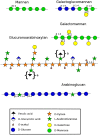
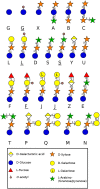
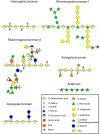
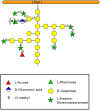
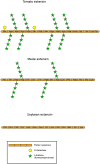
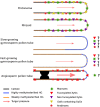
During evolution of land plants, the first colonizing species presented leafy-dominant gametophytes, found in non-vascular plants (bryophytes). Today, bryophytes include liverworts, mosses, and hornworts. In the first seedless vascular plants (lycophytes), the sporophytic stage of life started to be predominant. In the seed producing plants, gymnosperms and angiosperms , the gametophytic stage is restricted to reproduction. In mosses and ferns, the haploid spores germinate and form a protonema, which develops into a leafy gametophyte producing rhizoids for anchorage, water and nutrient uptakes. The basal gymnosperms (cycads and Ginkgo) reproduce by zooidogamy. Their pollen grains develop a multi-branched pollen tube that penetrates the nucellus and releases flagellated sperm cells that swim to the egg cell. The pollen grain of other gymnosperms (conifers and gnetophytes) as well as angiosperms germinates and produces a pollen tube that directly delivers the sperm cells to the ovule (siphonogamy). These different gametophytes, which are short or long-lived structures, share a common tip-growing mode of cell expansion. Tip-growth requires a massive cell wall deposition to promote cell elongation, but also a tight spatial and temporal control of the cell wall remodeling in order to modulate the mechanical properties of the cell wall. The growth rate of these cells is very variable depending on the structure and the species, ranging from very slow (protonemata, rhizoids, and some gymnosperm pollen tubes), to a slow to fast-growth in other gymnosperms and angiosperms. In addition, the structural diversity of the female counterparts in angiosperms (dry, semi-dry vs wet stigmas, short vs long, solid vs hollow styles) will impact the speed and efficiency of sperm delivery. As the evolution and diversity of the cell wall polysaccharides accompanied the diversification of cell wall structural proteins and remodeling enzymes, this review focuses on our current knowledge on the biochemistry, the distribution and remodeling of the main cell wall polymers (including cellulose, hemicelluloses, pectins, callose, arabinogalactan-proteins and extensins), during the tip-expansion of gametophytes from bryophytes, pteridophytes (lycophytes and monilophytes), gymnosperms and the monocot and eudicot angiosperms.
Keywords: cell-wall; evolution; gametophyte; land plants; pollen-tube; protonema; rhizoid; tip-growth.
Figures
References
-
- Abel W. O., Knebel W., Koop H. U., Marienfeld J. R., Quader H., Reski R., Schnepf E., Sprlein B. (1989). A cytokinin-sensitive mutant of the moss, Physcomitrella patens, defective in chloroplast division. Protoplasma 152, 1–13. 10.1007/BF01354234 - DOI
-
- Anderson J. R., Barnes W. S., Bedinger P. (2002). 2,6-Dichlorobenzonitrile, a cellulose biosynthesis inhibitor, affects morphology and structural integrity of petunia and lily pollen tubes. J. Plant Physiol. 159, 61–67. 10.1078/0176-1617-00651 - DOI
Publication types
LinkOut - more resources
Full Text Sources
Research Materials

