Secondary amines as coupling partners in direct catalytic asymmetric reductive amination
- PMID: 31057780
- PMCID: PMC6482873
- DOI: 10.1039/c9sc00323a
Secondary amines as coupling partners in direct catalytic asymmetric reductive amination
Abstract
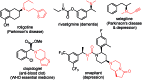
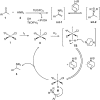
The secondary amine participating asymmetric reductive amination remains an unsolved problem in organic synthesis. Here we show for the first time that secondary amines are capable of effectively serving as N-sources in direct asymmetric reductive amination to afford corresponding tertiary chiral amines with the help of a selected additive set under mild conditions (0-25 °C). The applied chiral phosphoramidite ligands are readily prepared from BINOL and easily modified. Compared with common tertiary chiral amine synthetic methods, this procedure is much more concise and scalable, as exemplified by the facile synthesis of rivastigmine and N-methyl-1-phenylethanamine.
Figures
Similar articles
-
Iridium-catalyzed direct asymmetric reductive amination utilizing primary alkyl amines as the N-sources.Nat Commun. 2022 Jun 10;13(1):3344. doi: 10.1038/s41467-022-31045-5. Nat Commun. 2022. PMID: 35688909 Free PMC article.
-
Direct Asymmetric Reductive Amination for the Synthesis of (S)-Rivastigmine.Molecules. 2018 Aug 31;23(9):2207. doi: 10.3390/molecules23092207. Molecules. 2018. PMID: 30200331 Free PMC article.
-
An Iridium Catalytic System Compatible with Inorganic and Organic Nitrogen Sources for Dual Asymmetric Reductive Amination Reactions.Angew Chem Int Ed Engl. 2021 Dec 20;60(52):27307-27311. doi: 10.1002/anie.202112671. Epub 2021 Nov 16. Angew Chem Int Ed Engl. 2021. PMID: 34699113
-
[NAD(P)H-dependent oxidoreductases for synthesis of chiral amines by asymmetric reductive amination of ketones].Sheng Wu Gong Cheng Xue Bao. 2020 Sep 25;36(9):1794-1816. doi: 10.13345/j.cjb.190582. Sheng Wu Gong Cheng Xue Bao. 2020. PMID: 33164457 Review. Chinese.
-
NAD(P)H-Dependent Dehydrogenases for the Asymmetric Reductive Amination of Ketones: Structure, Mechanism, Evolution and Application.Adv Synth Catal. 2017 Jun 19;359(12):2011-2025. doi: 10.1002/adsc.201700356. Epub 2017 May 11. Adv Synth Catal. 2017. PMID: 30008635 Free PMC article. Review.
Cited by
-
Zinc Chloride-Catalyzed Synthesis of Carbamates: An Application for the Synthesis of the Anti-Alzheimer's Drug Rivastigmine.ACS Omega. 2022 Sep 26;7(40):36017-36027. doi: 10.1021/acsomega.2c05350. eCollection 2022 Oct 11. ACS Omega. 2022. PMID: 36249350 Free PMC article.
-
Multifunctional biocatalyst for conjugate reduction and reductive amination.Nature. 2022 Apr;604(7904):86-91. doi: 10.1038/s41586-022-04458-x. Epub 2022 Apr 6. Nature. 2022. PMID: 35388195
-
Structure-Based Design of Small Imine Reductase Panels for Target Substrates.ACS Catal. 2023 Sep 5;13(18):12310-12321. doi: 10.1021/acscatal.3c02278. eCollection 2023 Sep 15. ACS Catal. 2023. PMID: 37736118 Free PMC article.
-
The combination of asymmetric hydrogenation of olefins and direct reductive amination.Nat Commun. 2020 Jan 30;11(1):621. doi: 10.1038/s41467-020-14475-x. Nat Commun. 2020. PMID: 32001706 Free PMC article.
-
Iridium-catalyzed direct asymmetric reductive amination utilizing primary alkyl amines as the N-sources.Nat Commun. 2022 Jun 10;13(1):3344. doi: 10.1038/s41467-022-31045-5. Nat Commun. 2022. PMID: 35688909 Free PMC article.
References
-
- Jarvis L. M. Chem. Eng. News. 2016;94:12–17.
-
- Roughley S. D., Jordan A. M. J. Med. Chem. 2011;54:3451–3479. - PubMed
-
- Chiral Amine Synthesis: Methods, Developments and Applications, ed. T. C. Nugent, Wiley-VCH, Weinheim, 2010.
- Stereoselective Formation of Amines, Top. Curr. Chem., ed. W. Li and X. Zhang, 2014, p. 343. - PubMed
- Blakemore D. C., Castro L., Churcher I., Rees D. C., Thomas A. W., Wilson D. M., Wood A. Nat. Chem. 2018;10:383–394. - PubMed
-
- Nugent T. C., El-Shazly M. Adv. Synth. Catal. 2010;352:753–819.
- Xie J.-H., Zhu S.-F., Zhou Q.-L. Chem. Rev. 2011;111:1713–1760. - PubMed
- Xie J.-H., Zhu S.-F., Zhou Q.-L. Chem. Soc. Rev. 2012;41:4126–4139. - PubMed
- Ager D. J., de Vries A. H. M., de Vries J. G. Chem. Soc. Rev. 2012;41:3340–3380. - PubMed
-
- Magee M. P., Norton J. R. J. Am. Chem. Soc. 2001;123:1778–1779. - PubMed
- Ji Y., Feng G.-S., Chen M.-W., Shi L., Du H., Zhou Y.-G. Org. Chem. Front. 2017;4:1125–1129.
LinkOut - more resources
Full Text Sources
Other Literature Sources