250 kHz, 1.5 µm resolution SD-OCT for in-vivo cellular imaging of the human cornea
- PMID: 31065450
- PMCID: PMC6490998
- DOI: 10.1364/BOE.9.006569
250 kHz, 1.5 µm resolution SD-OCT for in-vivo cellular imaging of the human cornea
Abstract
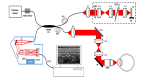
We present the first spectral domain optical coherence tomography (SD-OCT) system that combines an isotropic imaging resolution of ~1.5 µm in biological tissue with a 250 kHz image acquisition rate, for in vivo non-contact, volumetric imaging of the cellular structure of the human cornea. OCT images of the healthy human cornea acquired with this system reveal the cellular structure of the corneal epithelium, cellular debris and mucin clusters in the tear film, the shape, size and spatial distribution of the sub-basal corneal nerves and keratocytes in the corneal stroma, as well as reflections from endothelial nuclei. The corneal images presented here demonstrate the potential clinical value of the new high speed, high resolution OCT system for non-invasive diagnostics and monitoring the treatment of corneal diseases.
Conflict of interest statement
The authors declare that there are no conflicts of interest related to this article.
Figures





References
LinkOut - more resources
Full Text Sources
