Proteoglycans and Immunobiology of Cancer-Therapeutic Implications
- PMID: 31068944
- PMCID: PMC6491844
- DOI: 10.3389/fimmu.2019.00875
Proteoglycans and Immunobiology of Cancer-Therapeutic Implications
Abstract
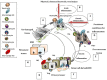
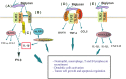
Disparity during the resolution of inflammation is closely related with the initiation and progression of the tumorigenesis. The transformed cells, through continuously evolving interactions, participate in various exchanges with the surrounding microenvironment consisting of extracellular matrix (ECM) components, cytokines embedded in the ECM, as well as the stromal cells. Proteoglycans (PGs), complex molecules consisting of a protein core into which one or more glycosaminoglycan (GAG) chains are covalently tethered, are important regulators of the cell/matrix interface and, consecutively, biological functions. The discrete expression of PGs and their interacting partners has been distinguished as specific for disease development in diverse cancer types. In this mini-review, we will critically discuss the roles of PGs in the complex processes of cancer-associated modulation of the immune response and analyze their mechanisms of action. A deeper understanding of mechanisms which are capable of regulating the immune response could be harnessed to treat malignant disease.
Keywords: cancer; extracellular matrix; immunobiology; proteoglycans; remodeling.
Figures
References
-
- Konsoulova A. Chapter 5. Principles of cancer immunobiology and immunotherapy of solid tumors. In: Metodiev E, editor. Immunopathology and Immunomodulation, INTECH; (2015).
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

