Effect of inoculum density on human-induced pluripotent stem cell expansion in 3D bioreactors
- PMID: 31069891
- PMCID: PMC6668975
- DOI: 10.1111/cpr.12604
Effect of inoculum density on human-induced pluripotent stem cell expansion in 3D bioreactors
Abstract
Objective: For optimized expansion of human-induced pluripotent stem cells (hiPSCs) with regards to clinical applications, we investigated the influence of the inoculum density on the expansion procedure in 3D hollow-fibre bioreactors.
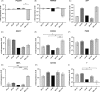
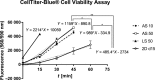
Materials and methods: Analytical-scale bioreactors with a cell compartment volume of 3 mL or a large-scale bioreactor with a cell compartment volume of 17 mL were used and inoculated with either 10 × 106 or 50 × 106 hiPSCs. Cells were cultured in bioreactors over 15 days; daily measurements of biochemical parameters were performed. At the end of the experiment, the CellTiter-Blue® Assay was used for culture activity evaluation and cell quantification. Also, cell compartment sections were removed for gene expression and immunohistochemistry analysis.
Results: The results revealed significantly higher values for cell metabolism, cell activity and cell yields when using the higher inoculation number, but also a more distinct differentiation. As large inoculation numbers require cost and time-extensive pre-expansion, low inoculation numbers may be used preferably for long-term expansion of hiPSCs. Expansion of hiPSCs in the large-scale bioreactor led to a successful production of 5.4 × 109 hiPSCs, thereby achieving sufficient cell amounts for clinical applications.
Conclusions: In conclusion, the results show a significant effect of the inoculum density on cell expansion, differentiation and production of hiPSCs, emphasizing the importance of the inoculum density for downstream applications of hiPSCs. Furthermore, the bioreactor technology was successfully applied for controlled and scalable production of hiPSCs for clinical use.
Keywords: 3D culture; bioreactor culture; cell expansion; human-induced pluripotent stem cells; inoculum density.
© 2019 The Authors. Cell Proliferation Published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors have no conflict of interest to declare.
Figures



References
-
- Schwartz SD, Regillo CD, Lam BL, et al. Human embryonic stem cell‐derived retinal pigment epithelium in patients with age‐related macular degeneration and Stargardt's macular dystrophy: follow‐up of two open‐label phase 1/2 studies. Lancet. 2015;385(9967):509‐516. 10.1016/S0140-6736(14)61376-3 - DOI - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

