Noise-induced hearing loss alters hippocampal glucocorticoid receptor expression in rats
- PMID: 31071644
- PMCID: PMC7035127
- DOI: 10.1016/j.heares.2019.04.013
Noise-induced hearing loss alters hippocampal glucocorticoid receptor expression in rats
Abstract
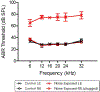
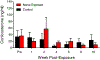
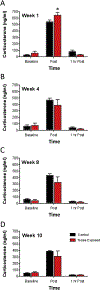
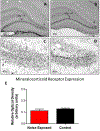
Although the effects of intense noise exposure on the peripheral and central auditory pathway have been well characterized, its effects on non-classical auditory structures in the brain, such as the hippocampus, are less well understood. Previously, we demonstrated that noise-induced hearing loss causes a significant long-term reduction in hippocampal neurogenesis and cell proliferation. Given the known suppressive effects of stress hormones on neurogenesis, the goal of the present study was to determine if activation of the stress response is an underlying mechanism for the long-term reduction in hippocampal neurogenesis observed following noise trauma. To accomplish this, we monitored basal and reactive blood plasma levels of the stress hormone corticosterone in rats for ten weeks following acoustic trauma, and quantified changes in hippocampal glucocorticoid and mineralocorticoid receptors. Our results indicate that long-term auditory deprivation does not cause a persistent increase in basal or reactive stress hormone levels in the weeks following noise exposure. Instead, we observed a greater decline in reactive corticosterone release in noise-exposed rats between the first and tenth week of sampling compared to control rats. We also observed a significant increase in hippocampal glucocorticoid receptor expression which may cause greater hippocampal sensitivity to circulating glucocorticoid levels and result in glucocorticoid-induced suppression of neurogenesis, as well as increased feedback inhibition on the HPA axis. No change in mineralocorticoid receptor expression was observed between control and noise exposed rats. These results highlight the adverse effect of intense noise exposure and auditory deprivation on the hippocampus.
Keywords: Corticosterone; Glucocorticoid receptors; Hearing loss; Mineralocorticoid receptors; Neurogenesis; Noise-exposure.
Copyright © 2019 Elsevier B.V. All rights reserved.
Conflict of interest statement
Conflict of Interest
The authors declare no conflict of interest.
Figures



References
-
- Altman J 1962. Are new neurons formed in the brains of adult mammals? Science 135:1127–1128. - PubMed
-
- Atkinson HC, Waddell BJ 1997. Circadian variation in basal plasma corticosterone and adrenocorticotropin in the rat: sexual dimorphism and changes across the estrous cycle. Endocrinology 138:3842–3848. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

