Tau covariance patterns in Alzheimer's disease patients match intrinsic connectivity networks in the healthy brain
- PMID: 31077982
- PMCID: PMC6510968
- DOI: 10.1016/j.nicl.2019.101848
Tau covariance patterns in Alzheimer's disease patients match intrinsic connectivity networks in the healthy brain
Abstract
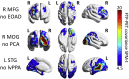
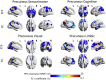
According to the network model of neurodegeneration, the spread of pathogenic proteins occurs selectively along connected brain regions. We tested in vivo whether the distribution of filamentous tau (measured with [18F]flortaucipir-PET), fibrillar amyloid-β ([11C]PIB-PET) and glucose hypometabolism ([18F]FDG-PET) follows the intrinsic functional organization of the healthy brain. We included 63 patients with Alzheimer's disease (AD; 30 male, 63 ± 8 years) who underwent [18F]flortaucipir, [11C]PIB and [18F]FDG PET, and 1000 young adults (427 male, 21 ± 3 years) who underwent task-free fMRI. We selected six predefined disease epicenters as seeds for whole-brain voxelwise covariance analyses to compare correlated patterns of tracer uptake across AD patients against fMRI intrinsic connectivity patterns in young adults. We found a striking convergence between [18F]flortaucipir covariance patterns and intrinsic connectivity maps (range Spearman rho's: 0.32-0.78, p < .001), which corresponded with expected functional networks (range goodness-of-fit: 3.8-8.2). The topography of amyloid-β covariance patterns was more diffuse and less network-specific, while glucose hypometabolic patterns were more spatially restricted than tau but overlapped with functional networks. These findings suggest that the spatial patterns of tau and glucose hypometabolism observed in AD resemble the functional organization of the healthy brain, supporting the notion that tau pathology spreads through circumscribed brain networks and drives neurodegeneration.
Keywords: Alzheimer's disease; Amyloid; Flortaucipir; Functional connectivity; PET; Tau.
Copyright © 2019. Published by Elsevier Inc.
Figures





References
-
- Albert M.S., DeKosky S.T., Dickson D., Dubois B., Feldman H.H., Fox N.C., Gamst A., Holtzman D.M., Jagust W.J., Petersen R.C., Snyder P.J., Carrillo M.C., Thies B., Phelps C.H. The diagnosis of mild cognitive impairment due to Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011;7:270–279. - PMC - PubMed
-
- Braak H., Braak E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 1991;82:239–259. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

