Clinical Evaluation of Human Papillomavirus Screening With p16/Ki-67 Dual Stain Triage in a Large Organized Cervical Cancer Screening Program
- PMID: 31081870
- PMCID: PMC6515572
- DOI: 10.1001/jamainternmed.2019.0306
Clinical Evaluation of Human Papillomavirus Screening With p16/Ki-67 Dual Stain Triage in a Large Organized Cervical Cancer Screening Program
Erratum in
-
Errors in Caption to Figure 2.JAMA Intern Med. 2019 Jul 1;179(7):1007. doi: 10.1001/jamainternmed.2019.2636. JAMA Intern Med. 2019. PMID: 31260006 Free PMC article. No abstract available.
Abstract
Importance: As cervical cancer screening transitions from Papanicolaou cytologic screening to primary human papillomavirus (HPV) testing worldwide, effective triage tests are needed to decide who among the HPV-positive women should receive further diagnostic evaluation to avoid unnecessary colposcopies and biopsies.
Objective: To evaluate the performance of the p16/Ki-67 dual stain (DS) and HPV16/18 genotyping for the triage of HPV-positive women.
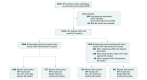
Design, setting, and participants: A prospective observational study was conducted within the cervical cancer screening program at Kaiser Permanente Northern California of 3225 HPV-positive women undergoing HPV and Papanicolaou cytologic testing with a valid DS result from September 16 to October 31, 2015, with follow-up through December 31, 2018.
Exposures: Human papillomavirus screening with partial genotyping and cytologic triage compared with DS triage.
Main outcomes and measures: Cervical intraepithelial neoplasia grade 3 or more severe (CIN3+) and grade 2 or more severe (CIN2+), diagnosed within 3 years after sample collection.
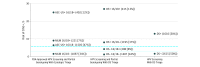
Results: A total of 3225 women (mean [SD] age, 37.9 [11.3] years) participated in the study. For triage of HPV-positive women with partial genotyping, DS showed better risk stratification for CIN3+ than did Papanicolaou cytologic testing, with women with positive DS results having a higher risk than women with positive Papanicolaou test results for CIN3+ (218 of 1818 [12.0%; 95% CI, 10.5%-13.5%] vs 219 of 2128 [10.3%; 95% CI, 9.0%-11.6%]; P = .005). Similarly, DS showed better risk stratification for CIN3+ compared with Papanicolaou cytologic testing in HPV-positive women, irrespective of genotyping. The greatest reassurance against CIN3+ was observed in HPV16/18-negative women with negative DS results, with a risk low enough to extend retesting intervals. Dual stain triage strategies required substantially fewer colposcopies per detection of CIN3+ compared with Papanicolaou cytologic testing, with a 32.1% (859 of 2677) reduction of colposcopies compared with the currently recommended triage strategy of HPV screening with Papanicolaou cytologic testing. Results for CIN2+ were very similar.
Conclusions and relevance: Triage of HPV-positive women with DS was superior to Papanicolaou cytologic testing in this study, demonstrating equal immediate detection of precancerous lesions and substantially reduced referral to colposcopy. These findings suggest that DS can safely replace Papanicolaou cytologic testing as a triage strategy for primary HPV screening, and that retesting intervals in HPV16/18-negative women with negative DS results can be safely extended to 3 years.
Conflict of interest statement
Figures
Comment in
-
Screening Options for Preventing Cervical Cancer.JAMA Intern Med. 2019 Jul 1;179(7):879-880. doi: 10.1001/jamainternmed.2019.0298. JAMA Intern Med. 2019. PMID: 31081850 No abstract available.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

