Selection of antimicrobial frog peptides and temporin-1DRa analogues for treatment of bacterial infections based on their cytotoxicity and differential activity against pathogens
- PMID: 31102497
- PMCID: PMC7891380
- DOI: 10.1111/cbdd.13569
Selection of antimicrobial frog peptides and temporin-1DRa analogues for treatment of bacterial infections based on their cytotoxicity and differential activity against pathogens
Abstract
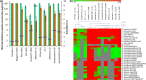
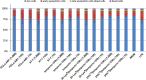
Cationic, amphipathic, α-helical host-defense peptides (HDPs) that are naturally secreted by certain species of frogs (Anura) possess potent broad-spectrum antimicrobial activity and show therapeutic potential as alternatives to treat infections by multidrug-resistant pathogens. Fourteen amphibian skin peptides and twelve analogues of temporin-1DRa were studied for their antimicrobial activities against clinically relevant human or animal skin infection-associated pathogens. For comparison, antimicrobial potencies of frog skin peptides against a range of probiotic lactobacilli were determined. We used the VITEK 2 system to define a profile of antibiotic susceptibility for the bacterial panel. The minimal inhibitory concentration (MIC) values of the naturally occurring temporin-1DRa, CPF-AM1, alyteserin-1c, hymenochirin-2B, and hymenochirin-4B for pathogenic bacteria were threefold to ninefold lower than the values for the tested probiotic strains. Similarly, temporin-1DRa and its [Lys4 ], [Lys5 ], and [Aib8 ] analogues showed fivefold to 6.5-fold greater potency against the pathogens. In the case of PGLa-AM1, XT-7, temporin-1DRa and its [D-Lys8 ] and [Aib13 ] analogues, no apoptosis or necrosis was detected in human peripheral blood mononuclear cells at concentrations below or above the MIC. Given the differential activity against commensal bacteria and pathogens, some of these peptides are promising candidates for further development into therapeutics for topical treatment of skin infections.
Keywords: HDP; antimicrobial peptide; biological screening.
© 2019 John Wiley & Sons A/S.
Conflict of interest statement
None declared.
Figures

Similar articles
-
Activities of four frog skin-derived antimicrobial peptides (temporin-1DRa, temporin-1Va and the melittin-related peptides AR-23 and RV-23) against anaerobic bacteria.Int J Antimicrob Agents. 2007 Mar;29(3):317-21. doi: 10.1016/j.ijantimicag.2006.09.007. Epub 2006 Dec 28. Int J Antimicrob Agents. 2007. PMID: 17196372
-
Strategies for transformation of naturally-occurring amphibian antimicrobial peptides into therapeutically valuable anti-infective agents.Methods. 2007 Aug;42(4):349-57. doi: 10.1016/j.ymeth.2007.01.004. Methods. 2007. PMID: 17560323
-
Effect of aminoisobutyric acid (Aib) substitutions on the antimicrobial and cytolytic activities of the frog skin peptide, temporin-1DRa.Peptides. 2007 Oct;28(10):2075-80. doi: 10.1016/j.peptides.2007.07.023. Epub 2007 Jul 27. Peptides. 2007. PMID: 17767978
-
Temporins: An Approach of Potential Pharmaceutic Candidates.Surg Infect (Larchmt). 2020 May;21(4):309-322. doi: 10.1089/sur.2019.266. Epub 2019 Dec 4. Surg Infect (Larchmt). 2020. PMID: 31804896 Review.
-
Antimicrobial peptides as a promising treatment option against Acinetobacter baumannii infections.Microb Pathog. 2020 Sep;146:104238. doi: 10.1016/j.micpath.2020.104238. Epub 2020 May 5. Microb Pathog. 2020. PMID: 32387392 Review.
Cited by
-
Discovery of a Novel Antimicrobial Peptide, Temporin-PKE, from the Skin Secretion of Pelophylax kl. esculentus, and Evaluation of Its Structure-Activity Relationships.Biomolecules. 2022 May 29;12(6):759. doi: 10.3390/biom12060759. Biomolecules. 2022. PMID: 35740884 Free PMC article.
-
A Novel Amphibian Antimicrobial Peptide, Phylloseptin-PV1, Exhibits Effective Anti-staphylococcal Activity Without Inducing Either Hepatic or Renal Toxicity in Mice.Front Microbiol. 2020 Oct 26;11:565158. doi: 10.3389/fmicb.2020.565158. eCollection 2020. Front Microbiol. 2020. PMID: 33193152 Free PMC article.
-
A Rapid In Vivo Toxicity Assessment Method for Antimicrobial Peptides.Toxics. 2024 May 25;12(6):387. doi: 10.3390/toxics12060387. Toxics. 2024. PMID: 38922067 Free PMC article.
References
-
- Afacan, N. J. , Yeung, A. T. , Pena, O. M. , & Hancock, R. E. (2012). Therapeutic potential of host defense peptides in antibiotic‐resistant infections. Current Pharmaceutical Design, 18, 807–819. - PubMed
-
- Al‐Ghaferi, N. , Kolodziejek, J. , Nowotny, N. , Coquet, L. , Jouenne, T. , Leprince, J. , … Conlon, J. M. (2010). Antimicrobial peptides from the skin secretions of the South‐East Asian frog Hylarana erythraea (Ranidae). Peptides, 31, 548–554. - PubMed
-
- Ali, M. F. , Soto, A. , Knoop, F. C. , & Conlon, J. M. (2001). Antimicrobial peptides isolated from skin secretions of the diploid frog, Xenopus tropicalis (Pipidae). Biochimica et Biophysica Acta, 1550, 81–89. - PubMed
-
- Bannoehr, J. , & Guardabassi, L. (2012). Staphylococcus pseudintermedius in the dog: Taxonomy, diagnostics, ecology, epidemiology and pathogenicity. Veterinary Dermatology, 23, 253–266, e51‐2. - PubMed
-
- Belkaid, Y. , & Tamoutounour, S. (2016). The influence of skin microorganisms on cutaneous immunity. Nature Reviews Immunology, 16, 353–366. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

