Impact of ROS-Induced Damage of TCA Cycle Enzymes on Metabolism and Virulence of Salmonella enterica serovar Typhimurium
- PMID: 31105651
- PMCID: PMC6491894
- DOI: 10.3389/fmicb.2019.00762
Impact of ROS-Induced Damage of TCA Cycle Enzymes on Metabolism and Virulence of Salmonella enterica serovar Typhimurium
Abstract
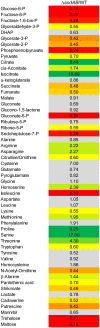
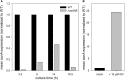
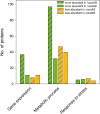
Salmonella enterica serovar Typhimurium (STM) is exposed to reactive oxygen species (ROS) originating from aerobic respiration, antibiotic treatment, and the oxidative burst occurring inside the Salmonella-containing vacuole (SCV) within host cells. ROS damage cellular compounds, thereby impairing bacterial viability and inducing cell death. Proteins containing iron-sulfur (Fe-S) clusters are particularly sensitive and become non-functional upon oxidation. Comprising five enzymes with Fe-S clusters, the TCA cycle is a pathway most sensitive toward ROS. To test the impact of ROS-mediated metabolic perturbations on bacterial physiology, we analyzed the proteomic and metabolic profile of STM deficient in both cytosolic superoxide dismutases (ΔsodAB). Incapable of detoxifying superoxide anions (SOA), endogenously generated SOA accumulate during growth. ΔsodAB showed reduced abundance of aconitases, leading to a metabolic profile similar to that of an aconitase-deficient strain (ΔacnAB). Furthermore, we determined a decreased expression of acnA in STM ΔsodAB. While intracellular proliferation in RAW264.7 macrophages and survival of methyl viologen treatment were not reduced for STM ΔacnAB, proteomic profiling revealed enhanced stress response. We conclude that ROS-mediated reduced expression and damage of aconitase does not impair bacterial viability or virulence, but might increase ROS amounts in STM, which reinforces the bactericidal effects of antibiotic treatment and immune responses of the host.
Keywords: aconitase; iron–sulfur cluster damage; metabolomics; oxidative stress; superoxide dismutase.
Figures




Similar articles
-
Characterization of Salmonella enterica serovar Typhimurium aconitase A.Microbiology (Reading). 2013 Jun;159(Pt 6):1209-1216. doi: 10.1099/mic.0.067934-0. Epub 2013 May 1. Microbiology (Reading). 2013. PMID: 23637460
-
Blocks in Tricarboxylic Acid Cycle of Salmonella enterica Cause Global Perturbation of Carbon Storage, Motility, and Host-Pathogen Interaction.mSphere. 2019 Dec 11;4(6):e00796-19. doi: 10.1128/mSphere.00796-19. mSphere. 2019. PMID: 31826974 Free PMC article.
-
Proteomics of intracellular Salmonella enterica reveals roles of Salmonella pathogenicity island 2 in metabolism and antioxidant defense.PLoS Pathog. 2019 Apr 22;15(4):e1007741. doi: 10.1371/journal.ppat.1007741. eCollection 2019 Apr. PLoS Pathog. 2019. PMID: 31009521 Free PMC article.
-
Aconitases: Non-redox Iron-Sulfur Proteins Sensitive to Reactive Species.Acc Chem Res. 2019 Sep 17;52(9):2609-2619. doi: 10.1021/acs.accounts.9b00150. Epub 2019 Jul 9. Acc Chem Res. 2019. PMID: 31287291 Review.
-
Superoxide-driven aconitase FE-S center cycling.Biosci Rep. 1997 Feb;17(1):33-42. doi: 10.1023/a:1027383100936. Biosci Rep. 1997. PMID: 9171919 Review.
Cited by
-
Preovulatory follicular fluid and serum metabolome profiles in lactating beef cows with thin, moderate, and obese body condition.J Anim Sci. 2022 Jul 1;100(7):skac152. doi: 10.1093/jas/skac152. J Anim Sci. 2022. PMID: 35772755 Free PMC article.
-
Protein Purification and Digestion Methods for Bacterial Proteomic Analyses.Methods Mol Biol. 2022;2456:63-70. doi: 10.1007/978-1-0716-2124-0_5. Methods Mol Biol. 2022. PMID: 35612735
-
Mitochondrial aconitase 1 regulates age-related memory impairment via autophagy/mitophagy-mediated neural plasticity in middle-aged flies.Aging Cell. 2021 Dec;20(12):e13520. doi: 10.1111/acel.13520. Epub 2021 Nov 19. Aging Cell. 2021. PMID: 34799973 Free PMC article.
-
Analysis of Regulatory Mechanism of AcrB and CpxR on Colistin Susceptibility Based on Transcriptome and Metabolome of Salmonella Typhimurium.Microbiol Spectr. 2023 Aug 17;11(4):e0053023. doi: 10.1128/spectrum.00530-23. Epub 2023 Jun 26. Microbiol Spectr. 2023. PMID: 37358428 Free PMC article.
-
Reactive Oxygen Species: From Tumorigenesis to Therapeutic Strategies in Cancer.Cancer Med. 2025 May;14(10):e70947. doi: 10.1002/cam4.70947. Cancer Med. 2025. PMID: 40377005 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

