Mitochondrial complex III is necessary for endothelial cell proliferation during angiogenesis
- PMID: 31106291
- PMCID: PMC6521885
- DOI: 10.1038/s42255-018-0011-x
Mitochondrial complex III is necessary for endothelial cell proliferation during angiogenesis
Abstract
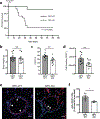
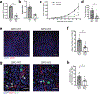
Endothelial cells (ECs) require glycolysis for proliferation and migration during angiogenesis; however, the necessity for the mitochondrial respiratory chain during angiogenesis is not known. Here we report that inhibition of respiratory chain complex III impairs proliferation, but not migration of ECs in vitro by decreasing the NAD+/NADH ratio. To determine whether mitochondrial respiration is necessary for angiogenesis in vivo, we conditionally ablate a subunit of the respiratory chain complex III (QPC) in ECs. Loss of QPC decreases respiration, resulting in diminished EC proliferation, and impairment in retinal and tumor angiogenesis. Loss of QPC does not decrease genes associated with anabolism or nucleotides levels in ECs, but diminishes amino acid levels. Our findings indicate that mitochondrial respiration is necessary for angiogenesis, and that the primary role of mitochondria in ECs is to serve as biosynthetic organelles for cell proliferation.
Figures





Similar articles
-
The role of glycolysis and mitochondrial respiration in the formation and functioning of endothelial tip cells during angiogenesis.Sci Rep. 2019 Aug 30;9(1):12608. doi: 10.1038/s41598-019-48676-2. Sci Rep. 2019. PMID: 31471554 Free PMC article.
-
The mitochondrial permeability transition pore regulates endothelial bioenergetics and angiogenesis.Circ Res. 2015 Apr 10;116(8):1336-45. doi: 10.1161/CIRCRESAHA.116.304881. Epub 2015 Feb 26. Circ Res. 2015. PMID: 25722455 Free PMC article.
-
Endothelial specific SIRT3 deletion impairs glycolysis and angiogenesis and causes diastolic dysfunction.J Mol Cell Cardiol. 2017 Nov;112:104-113. doi: 10.1016/j.yjmcc.2017.09.007. Epub 2017 Sep 19. J Mol Cell Cardiol. 2017. PMID: 28935506 Free PMC article.
-
Mitochondria in endothelial cells: Sensors and integrators of environmental cues.Redox Biol. 2017 Aug;12:821-827. doi: 10.1016/j.redox.2017.04.021. Epub 2017 Apr 18. Redox Biol. 2017. PMID: 28448943 Free PMC article. Review.
-
Mitochondria in endothelial cells angiogenesis and function: current understanding and future perspectives.J Transl Med. 2023 Jul 5;21(1):441. doi: 10.1186/s12967-023-04286-1. J Transl Med. 2023. PMID: 37407961 Free PMC article. Review.
Cited by
-
Mitochondria on the move: Horizontal mitochondrial transfer in disease and health.J Cell Biol. 2023 Mar 6;222(3):e202211044. doi: 10.1083/jcb.202211044. Epub 2023 Feb 16. J Cell Biol. 2023. PMID: 36795453 Free PMC article.
-
Common methods in mitochondrial research (Review).Int J Mol Med. 2022 Oct;50(4):126. doi: 10.3892/ijmm.2022.5182. Epub 2022 Aug 25. Int J Mol Med. 2022. PMID: 36004457 Free PMC article.
-
Resistance Mechanisms of the Metastatic Tumor Microenvironment to Anti-Angiogenic Therapy.Front Oncol. 2022 May 19;12:897927. doi: 10.3389/fonc.2022.897927. eCollection 2022. Front Oncol. 2022. PMID: 35664794 Free PMC article. Review.
-
Blocking cholesterol formation and turnover improves cellular and mitochondria function in murine heart microvascular endothelial cells and cardiomyocytes.Front Physiol. 2023 Sep 7;14:1216267. doi: 10.3389/fphys.2023.1216267. eCollection 2023. Front Physiol. 2023. PMID: 37745244 Free PMC article.
-
Mitochondria complex III-generated superoxide is essential for IL-10 secretion in macrophages.Sci Adv. 2025 Jan 24;11(4):eadu4369. doi: 10.1126/sciadv.adu4369. Epub 2025 Jan 22. Sci Adv. 2025. PMID: 39841842 Free PMC article.
References
-
- De Bock K et al. Role of PFKFB3-driven glycolysis in vessel sprouting. Cell 154, 651–663 (2013). - PubMed
-
- Yeh W-L, Lin C-J & Fu W-M Enhancement of glucose transporter expression of brain endothelial cells by vascular endothelial growth factor derived from glioma exposed to hypoxia. Mol. Pharmacol. 73, 170–177 (2008). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

