Identification of novel compound heterozygous mutations in ACO2 in a patient with progressive cerebral and cerebellar atrophy
- PMID: 31106992
- PMCID: PMC6625133
- DOI: 10.1002/mgg3.698
Identification of novel compound heterozygous mutations in ACO2 in a patient with progressive cerebral and cerebellar atrophy
Abstract
Background: The tricarboxylic acid (TCA) cycle is a sequence of catabolic reactions within the mitochondrial matrix, and is a central pathway for cellular energy metabolism. Genetic defects affecting the TCA cycle are known to cause severe multisystem disorders.
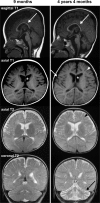
Methods: We performed whole exome sequencing of genomic DNA of a patient with progressive cerebellar and cerebral atrophy, hypotonia, ataxia, seizure disorder, developmental delay, ophthalmological abnormalities and hearing loss. We also performed biochemical studies using patient fibroblasts.
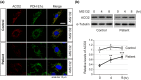
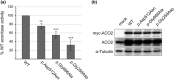
Results: We identified new compound heterozygous mutations (c.1534G > A, p.Asp512Asn and c.1997G > C, p.Gly666Ala) in ACO2, which encodes aconitase 2, a component of the TCA cycle. In patient fibroblasts, the aconitase activity was reduced to 15% of that of the control, and the aconitase 2 level decreased to 36% of that of the control. As such a decrease in aconitase 2 in patient fibroblasts was partially restored by proteasome inhibition, mutant aconitase 2 was suggested to be relatively unstable and rapidly degraded after being synthesized. In addition, the activity of the father-derived variant of aconitase 2 (p.Gly666Ala), which had a mutation near the active center, was 55% of that of wild-type.
Conclusion: The marked reduction of aconitase activity in patient fibroblasts was due to the combination of decreased aconitase 2 amount and activity due to mutations. Reduced aconitase activity directly suppresses the TCA cycle, resulting in mitochondrial dysfunction, which may lead to symptoms similar to those observed in mitochondrial diseases.
Keywords: ACO2; TCA cycle; aconitase; ataxia; brain atrophy; hypotonia; mitochondria.
© 2019 The Authors. Molecular Genetics & Genomic Medicine published by Wiley Periodicals, Inc.
Conflict of interest statement
None.
Figures

References
-
- Bourgeron, T. , Chretien, D. , Poggi-Bach, J. , Doonan, S. , Rabier, D. , Letouzé, P. , … Rustin, P. (1994). Mutation of the fumarase gene in two siblings with progressive encephalopathy and fumarase deficiency. Journal of Clinical Investigation, 93(6), 2514–2518. 10.1172/JCI117261. - DOI - PMC - PubMed
-
- Elpeleg, O. , Miller, C. , Hershkovitz, E. , Bitner‐Glindzicz, M. , Bondi‐Rubinstein, G. , Rahman, S. , … Saada, A. (2005). Deficiency of the ADP‐forming succinyl‐CoA synthase activity is associated with encephalomyopathy and mitochondrial DNA depletion. The American Journal of Human Genetics, 76(6), 1081–1086. 10.1086/430843 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

