Performance of 2 Commercial Serologic Tests for Diagnosing Zika Virus Infection
- PMID: 31107211
- PMCID: PMC6537740
- DOI: 10.3201/eid2506.180361
Performance of 2 Commercial Serologic Tests for Diagnosing Zika Virus Infection
Abstract
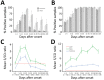
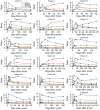
Reliable serologic tests are needed for diagnosis and surveillance of Zika virus infection. We evaluated the Euroimmun and Dia.Pro serologic tests for detection of Zika virus IgM and IgG by using a panel of 199 samples from a region endemic for flaviviruses. Kinetics of Zika virus antibodies were monitored from 300 sequential specimens sampled over a period of 10 months after infection. We observed suboptimal performance; sensitivity for Zika virus IgM was low, especially in the Euroimmun assay (49%), whereas IgM could be detected for months with the Dia.pro assay. The specificity of the Zika virus IgG assays was also low, especially that of Dia.Pro (62%); findings were strongly influenced by the epidemiologic context. These results highlight the complexity of serologic diagnosis of Zika virus infection in regions endemic for flaviviruses. Accurate analysis of the performance of assays is required to adapt and interpret algorithms.
Keywords: French Guiana; Zika virus; antibody kinetics; cross reactions; diagnosis; sensitivity; serology; specificity; viruses.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

