The Blood Pressure Pendulum following Spinal Cord Injury: Implications for Vascular Cognitive Impairment
- PMID: 31109053
- PMCID: PMC6567094
- DOI: 10.3390/ijms20102464
The Blood Pressure Pendulum following Spinal Cord Injury: Implications for Vascular Cognitive Impairment
Abstract
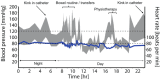
Cognitive impairment following spinal cord injury (SCI) has received considerable attention in recent years. Among the various systemic effects of SCI that contribute towards cognitive decline in this population, cardiovascular dysfunction is arguably one of the most significant. The majority of individuals with a cervical or upper-thoracic SCI commonly experience conditions called orthostatic hypotension and autonomic dysreflexia, which are characterized by dangerous fluctuations in systemic blood pressure (BP). Herein, we review the potential impact of extreme BP lability on vascular cognitive impairment (VCI) in individuals with SCI. Albeit preliminary in the SCI population, there is convincing evidence that chronic hypotension and hypertension in able-bodied individuals results in devastating impairments in cerebrovascular health, leading to VCI. We discuss the pertinent literature, and while drawing mechanistic comparisons between able-bodied cohorts and individuals with SCI, we emphasize the need for additional research to elucidate the mechanisms of cognitive impairment specific to the SCI population. Lastly, we highlight the current and potential future therapies to manage and treat BP instability, thereby possibly mitigating VCI in the SCI population.
Keywords: autonomic dysreflexia; cerebrovascular health; orthostatic hypotension; spinal cord injury; vascular cognitive impairment.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures

Similar articles
-
Contemporary Cardiovascular Concerns after Spinal Cord Injury: Mechanisms, Maladaptations, and Management.J Neurotrauma. 2015 Dec 15;32(24):1927-42. doi: 10.1089/neu.2015.3903. Epub 2015 Sep 1. J Neurotrauma. 2015. PMID: 25962761 Review.
-
Diverse cognitive impairment after spinal cord injury is associated with orthostatic hypotension symptom burden.Physiol Behav. 2020 Jan 1;213:112742. doi: 10.1016/j.physbeh.2019.112742. Epub 2019 Nov 16. Physiol Behav. 2020. PMID: 31738949
-
Autonomic dysreflexia: current evidence related to unstable arterial blood pressure control among athletes with spinal cord injury.Clin J Sport Med. 2012 Jan;22(1):39-45. doi: 10.1097/JSM.0b013e3182420699. Clin J Sport Med. 2012. PMID: 22222591 Review.
-
Vascular dysfunctions following spinal cord injury.J Med Life. 2010 Jul-Sep;3(3):275-85. J Med Life. 2010. PMID: 20945818 Free PMC article. Review.
-
Spinal cord injury medicine. 2. Medical complications after spinal cord injury: Identification and management.Arch Phys Med Rehabil. 2002 Mar;83(3 Suppl 1):S58-64, S90-8. doi: 10.1053/apmr.2002.32159. Arch Phys Med Rehabil. 2002. PMID: 11973698 Review.
Cited by
-
Contributing Factors to Endothelial Dysfunction in Individuals with Spinal Cord Injuries.Pulse (Basel). 2024 May 7;12(1):49-57. doi: 10.1159/000539199. eCollection 2024 Jan-Dec. Pulse (Basel). 2024. PMID: 39022560 Free PMC article. Review.
-
A prospective cohort study investigating contributors to mild cognitive impairment in adults with spinal cord injury: study protocol.BMC Neurol. 2020 Sep 11;20(1):341. doi: 10.1186/s12883-020-01899-7. BMC Neurol. 2020. PMID: 32917161 Free PMC article.
-
The Role of Hydrogen Sulfide in Regulation of Cell Death following Neurotrauma and Related Neurodegenerative and Psychiatric Diseases.Int J Mol Sci. 2023 Jun 28;24(13):10742. doi: 10.3390/ijms241310742. Int J Mol Sci. 2023. PMID: 37445920 Free PMC article. Review.
-
Cardiovascular safety of transcutaneous spinal cord stimulation in cervical spinal cord injury.Neurotherapeutics. 2025 Mar;22(2):e00528. doi: 10.1016/j.neurot.2025.e00528. Epub 2025 Jan 31. Neurotherapeutics. 2025. PMID: 39893085 Free PMC article.
-
Cumulative Transcutaneous Spinal Stimulation with Locomotor Training Safely Improves Trunk Control in Children with Spinal Cord Injury: Pilot Study.Children (Basel). 2025 Jun 21;12(7):817. doi: 10.3390/children12070817. Children (Basel). 2025. PMID: 40723009 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

