R-spondin2 promotes hematopoietic differentiation of human pluripotent stem cells by activating TGF beta signaling
- PMID: 31109354
- PMCID: PMC6528258
- DOI: 10.1186/s13287-019-1242-9
R-spondin2 promotes hematopoietic differentiation of human pluripotent stem cells by activating TGF beta signaling
Abstract
Background: Human pluripotent stem cells (hPSCs) provide supplies of potential functional blood cells to suffice the clinical needs. However, the underlying mechanism of generating genuine hematopoietic stem cells (HSCs) and functional blood cells from hPSCs remains largely elusive.
Method: In this study, we supplied R-spondin2 exogenously during hematopoietic differentiation of hPSCs under various culture conditions and analyzed the production of hematopoietic progenitor cells (HPCs). We further added R-spondin2 at different temporal window to pin down the stage at which R-spondin2 conferred its effects. RNA-SEQ-based gene profiling was applied to analyze genes with significantly altered expression and altered signaling pathways. Finally, megakaryocytic differentiation and platelet generation were determined using HPCs with R-spondin2 treatment.
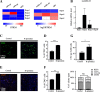
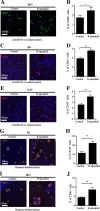
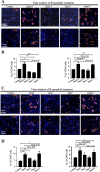
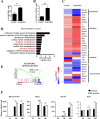
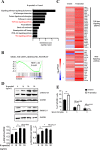
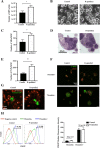
Results: We found that R-spondin2 generated by hematopoiesis-supporting stromal cells significantly enhances hematopoietic differentiation of hPSCs. Supply of R-spondin2 exogenously at the early stage of mesoderm differentiation elevates the generation of APLNR+ cells. Furthermore, early treatment of cells with R-spondin2 enables us to increase the output of hPSC-derived platelet-like particles (PLPs) with intact function. At the mechanistic level, R-spondin2 activates TGF-β signaling to promote the hematopoietic differentiation.
Conclusions: Our results demonstrate that a transient supply of R-spondin2 can efficiently promote hematopoietic development by activating both WNT and TGF-β signaling. R-spondin2 can be therefore used as a powerful tool for large-scale generation of functional hematopoietic progenitors and platelets for translational medicine.
Keywords: Hematopoietic differentiation; Human pluripotent stem cells; Mesoderm; R-spondin2; TGF-β signaling.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Similar articles
-
The balance of positive and negative effects of TGF-β signaling regulates the development of hematopoietic and endothelial progenitors in human pluripotent stem cells.Stem Cells Dev. 2013 Oct 15;22(20):2765-76. doi: 10.1089/scd.2013.0008. Epub 2013 Jul 19. Stem Cells Dev. 2013. PMID: 23758278 Free PMC article.
-
LGR4, Not LGR5, Enhances hPSC Hematopoiesis by Facilitating Mesoderm Induction via TGF-Beta Signaling Activation.Cell Rep. 2020 May 5;31(5):107600. doi: 10.1016/j.celrep.2020.107600. Cell Rep. 2020. PMID: 32375050
-
Cooperative Effect of Erythropoietin and TGF-β Inhibition on Erythroid Development in Human Pluripotent Stem Cells.J Cell Biochem. 2015 Dec;116(12):2735-43. doi: 10.1002/jcb.25233. J Cell Biochem. 2015. PMID: 26012423
-
Progress and challenges in generating functional hematopoietic stem/progenitor cells from human pluripotent stem cells.Cytotherapy. 2015 Apr;17(4):344-58. doi: 10.1016/j.jcyt.2015.01.003. Epub 2015 Feb 10. Cytotherapy. 2015. PMID: 25680303 Review.
-
Development of hematopoietic stem and progenitor cells from human pluripotent stem cells.J Cell Biochem. 2015 Jul;116(7):1179-89. doi: 10.1002/jcb.25097. J Cell Biochem. 2015. PMID: 25740540 Free PMC article. Review.
Cited by
-
De Novo Generation of Human Hematopoietic Stem Cells from Pluripotent Stem Cells for Cellular Therapy.Cells. 2023 Jan 14;12(2):321. doi: 10.3390/cells12020321. Cells. 2023. PMID: 36672255 Free PMC article. Review.
-
Widespread presence of bone marrow-like hematopoietic stem cell niche in invertebrate skeletons.Sci Adv. 2025 Jun 27;11(26):eadw0958. doi: 10.1126/sciadv.adw0958. Epub 2025 Jun 25. Sci Adv. 2025. PMID: 40561015 Free PMC article.
-
The biological functions and related signaling pathways of SPON2.Front Oncol. 2024 Jan 9;13:1323744. doi: 10.3389/fonc.2023.1323744. eCollection 2023. Front Oncol. 2024. PMID: 38264743 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

