Comparison of pegylated interferon monotherapy and de novo pegylated interferon plus tenofovir combination therapy in patients with chronic hepatitis B
- PMID: 31114265
- PMCID: PMC6489617
- DOI: 10.2147/IDR.S195144
Comparison of pegylated interferon monotherapy and de novo pegylated interferon plus tenofovir combination therapy in patients with chronic hepatitis B
Abstract
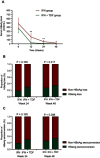
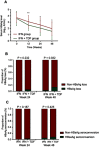
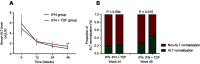
Background and aim: We aimed to evaluate the effectiveness of pegylated interferon (Peg-IFN) monotherapy (IFN group) and combination therapy with tenofovir (TDF) and Peg-IFN (IFN+TDF group) in chronic hepatitis B (CHB) patients. Patients and methods: Data of 143 CHB patients were analyzed in this study. All patients enrolled received liver biopsy. Virologic responses were defined as hepatitis B virus (HBV) DNA <100 IU/mL, biochemical responses were defined as normalization of alanine aminotransferse (ALT) levels, and HBeAg serological response was defined as HBeAg loss or HBeAg seroconversion to HBeAb. HBsAg serological response was defined as HBsAg loss or HBsAg seroconversion to HBsAb. Results: We observed that a total of 16.7% (11/66) and 33.8% (26/77) patients in IFN and IFN+TDF group achieved complete viral suppression after 48 weeks treatment (P=0.02). Although HBeAg levels in CHB patients in the IFN+TDF group decreased more rapidly during the 48-week treatment, we did not observe significant differences in HBeAg serological loss or seroconversion rates between the two groups at 24 and 48 weeks. HBsAg loss was observed in 13.0% (10/77) of CHB patients in the IFN+TDF group at 48 weeks, compared with only 3% (2/66) patients in the IFN group (P=0.032). No significant difference was observed in HBsAg seroconversion rate between the two groups during 48-week treatment. The biochemical response rate was also significantly higher in the IFN+TDF group than that in the IFN group at week 48 (P=0.015). Multivariate logistic analysis showed that IFN+TDF treatment (OR=4.41, P=0.003), severe baseline hepatic inflammation (OR=4.16, P<0.001), and lower baseline serum HBV DNA levels (OR=0.98, P=0.03) were strong predictors for the virological response. Younger age (OR=0.89, P=0.01), higher baseline ALT level (OR=1.01, P=0.038), and lower baseline HBeAg level (OR=0.99, P=0.008) were independent predictors for HBeAg sero-response after 48 weeks treatment. While only severe liver fibrosis (OR=1.69, P=0.028) and lower baseline HBsAg level (OR=0.22, P=0.005) were independent factors associated with HBsAg sero-response after 48 weeks treatment. Conclusion: Peg-IFN combined with TDF may increase the virological response rate, biochemical response rate, and HBsAg loss rate in patients with CHB infection. The combination treatment is more suitable for those patients who are likely to respond to the treatment.
Keywords: hepatitis B; pegylated interferon; tenofovir; virological response.
Conflict of interest statement
The authors declare that they have no financial or personal relationships with other people or organizations that could inappropriately influence this work. The authors report no other conflicts of interest in this work.
Figures

Similar articles
-
Effect of Pegylated Interferon Plus Tenofovir Combination on Higher Hepatitis B Surface Antigen Loss in Treatment-naive Patients With Hepatitis B e Antigen -positive Chronic Hepatitis B: A Real-world Experience.Clin Ther. 2021 Mar;43(3):572-581.e3. doi: 10.1016/j.clinthera.2020.12.022. Epub 2021 Jan 27. Clin Ther. 2021. PMID: 33516527
-
Comparative efficacy and safety of pegylated interferon-alpha monotherapy vs combination therapies with entecavir or tenofovir in chronic hepatitis B patients.Microbiol Spectr. 2025 Apr 2;13(5):e0269424. doi: 10.1128/spectrum.02694-24. Online ahead of print. Microbiol Spectr. 2025. PMID: 40172187 Free PMC article.
-
Peg-interferon alpha add-on Tenofovir disoproxil fumarate achieved more HBsAg loss in HBeAg-positive chronic hepatitis B naïve patients.J Viral Hepat. 2021 Oct;28(10):1381-1391. doi: 10.1111/jvh.13571. Epub 2021 Jul 14. J Viral Hepat. 2021. PMID: 34228855 Clinical Trial.
-
Treatment of chronic hepatitis B: case selection and duration of therapy.J Gastroenterol Hepatol. 2002 Apr;17(4):409-14. doi: 10.1046/j.1440-1746.2002.02767.x. J Gastroenterol Hepatol. 2002. PMID: 11982721 Review.
-
Optimal therapy of chronic hepatitis B: how do I treat my HBeAg-negative patients?Liver Int. 2015 Jan;35 Suppl 1:107-13. doi: 10.1111/liv.12717. Liver Int. 2015. PMID: 25529095 Review.
Cited by
-
Dynamic change of depression and anxiety after chemotherapy among patients with ovarian cancer.Medicine (Baltimore). 2019 Aug;98(31):e16620. doi: 10.1097/MD.0000000000016620. Medicine (Baltimore). 2019. PMID: 31374028 Free PMC article.
-
Risk factors associated with hepatic osteopathy in HBV related cirrhosis measured by liver stiffness: An Observational study.Medicine (Baltimore). 2019 Aug;98(31):e16628. doi: 10.1097/MD.0000000000016628. Medicine (Baltimore). 2019. PMID: 31374030 Free PMC article.
-
Comparison of clinical features and outcomes between HBV-related and non-B non-C hepatocellular carcinoma.Infect Agent Cancer. 2020 Feb 14;15:11. doi: 10.1186/s13027-020-0273-2. eCollection 2020. Infect Agent Cancer. 2020. PMID: 32082414 Free PMC article.
-
Smoking behavior and smoking index as prognostic indicators for patients with esophageal squamous cell carcinoma who underwent surgery: A large cohort study in Guangzhou, China.Tob Induc Dis. 2020 Feb 12;18:9. doi: 10.18332/tid/117428. eCollection 2020. Tob Induc Dis. 2020. PMID: 32071596 Free PMC article.
-
Increased expression of protease-activated receptors 2 indicates poor prognosis in HBV related hepatocellular carcinoma.Infect Agent Cancer. 2019 Nov 21;14:39. doi: 10.1186/s13027-019-0256-3. eCollection 2019. Infect Agent Cancer. 2019. PMID: 31768188 Free PMC article.
References
-
- Cai S, Cao J, Yu T, Xia M, Peng J. Effectiveness of entecavir or telbivudine therapy in patients with chronic hepatitis B virus infection pre-treated with interferon compared with de novo therapy with entecavir and telbivudine. Medicine (Baltimore). 2017;96:e7021. doi:10.1097/MD.0000000000007021 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

