Cell non-autonomous functions of S100a4 drive fibrotic tendon healing
- PMID: 31124787
- PMCID: PMC6546390
- DOI: 10.7554/eLife.45342
Cell non-autonomous functions of S100a4 drive fibrotic tendon healing
Abstract
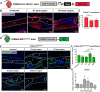
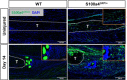
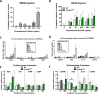
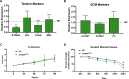
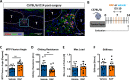
Identification of pro-regenerative approaches to improve tendon healing is critically important as the fibrotic healing response impairs physical function. In the present study we tested the hypothesis that S100a4 haploinsufficiency or inhibition of S100a4 signaling improves tendon function following acute injury and surgical repair in a murine model. We demonstrate that S100a4 drives fibrotic tendon healing primarily through a cell non-autonomous process, with S100a4 haploinsufficiency promoting regenerative tendon healing. Moreover, inhibition of S100a4 signaling via antagonism of its putative receptor, RAGE, also decreases scar formation. Mechanistically, S100a4 haploinsufficiency decreases myofibroblast and macrophage content at the site of injury, with both cell populations being key drivers of fibrotic progression. Moreover, S100a4-lineage cells become α-SMA+ myofibroblasts, via loss of S100a4 expression. Using a combination of genetic mouse models, small molecule inhibitors and in vitro studies we have defined S100a4 as a novel, promising therapeutic candidate to improve tendon function after acute injury.
Keywords: RAGE; S100a4; fibrosis; mouse; regeneration; regenerative medicine; stem cells; tendon.
© 2019, Ackerman et al.
Conflict of interest statement
JA, AN, VS, KB, EK, AL No competing interests declared
Figures


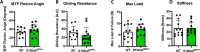




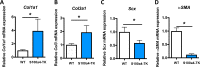




References
-
- Aydin A, Topalan M, Mezdeği A, Sezer I, Ozkan T, Erer M, Ozkan S. Single-stage flexor tendoplasty in the treatment of flexor tendon injuries. Acta Orthopaedica Et Traumatologica Turcica. 2004;38:54–59. - PubMed
-
- Belot N, Pochet R, Heizmann CW, Kiss R, Decaestecker C. Extracellular S100A4 stimulates the migration rate of astrocytic tumor cells by modifying the organization of their actin cytoskeleton. Biochimica Et Biophysica Acta (BBA) - Proteins and Proteomics. 2002;1600:74–83. doi: 10.1016/S1570-9639(02)00447-8. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

