One-year follow-up of a remotely delivered epilepsy self-management program in high-risk people with epilepsy
- PMID: 31126825
- PMCID: PMC7370541
- DOI: 10.1016/j.yebeh.2019.04.034
One-year follow-up of a remotely delivered epilepsy self-management program in high-risk people with epilepsy
Abstract
Objective: "Self-management for people with epilepsy and a history of negative health events" (SMART) is a novel group-format epilepsy self-management intervention demonstrated to reduce negative health events (NHEs) such as accidents, emergency department visits, and seizures in adults with epilepsy in a 6-month prospective randomized controlled trial (RCT); SMART also reduced depressive symptoms and improved health functioning and quality of life. This report describes the longer-term (12-month) post-efficacy RCT outcomes in adults with epilepsy who received SMART.
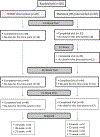
Methods: After completing a 6-month, prospective RCT that demonstrated efficacy of SMART vs 6-month waitlist control (WL), adults ≥18 years of age with epilepsy were followed for an additional 12 months. Individuals originally randomized to WL received the 8-week SMART intervention immediately following the conclusion of the RCT. For this long-term extension analysis, assessments were conducted at 24 weeks (the 6-month primary outcome time-point of the efficacy RCT), at 32 weeks for individuals originally randomized to WL, and at 48 weeks and 72 weeks for all individuals. Outcomes assessed included past 6-month NHE counts, depressive symptoms assessed with the 9-item Patient Health Questionnaire (PHQ-9) and Montgomery-Asberg Depression Rating Scale (MADRS), and quality of life assessed with the 10-item Quality of Life in Epilepsy (QOLIE-10).
Results: At the beginning of this long-term observational period (24-week follow-up time point for the original RCT), there were 50 individuals in the group originally randomized to SMART and 52 originally randomized to WL. Mean age was 41.4 years, 70% women (N = 71), 64% (N = 65) African-American, and 8% Hispanic (N = 8). Study attrition from week 24 to week 72 was 8% in the arm originally randomized to SMART and 17% in the arm originally randomized to WL. During the 12-month observation period (24 weeks to 72 weeks), there were a total of 44 serious adverse events and 4 deaths, none related to study participation. There was no significant change in total past 6-month NHE counts in the group originally randomized to SMART, although the group had significantly reduced 6-month seizure counts. The group originally randomized to WL, who received SMART during this observational period, had a reduction in total NHE counts. The group originally randomized to SMART had relatively stable levels on other outcome variables except for a trend for improved MADRS (p = 0.08). In the group originally randomized to WL, there were significant improvements in PHQ-9 (p = 0.01), MADRS (p ≤ 0.01), and QOLIE-10 (p = 0.004).
Conclusions: This post-RCT extension study suggests that adults with epilepsy who participate in the SMART intervention sustain clinical effects at 1-year follow-up and may have incremental improvements in seizure frequency and mood. Future research needs to identify opportunities for scale-up and outreach to other high-risk groups with epilepsy.
Keywords: Depression; Epilepsy; Quality of life; Seizures; Self-management.
Copyright © 2019 Elsevier Inc. All rights reserved.
Conflict of interest statement
Disclosure of Conflicts of Interest
Dr. Sajatovic has research grants from Otsuka, Alkermes, Merck, Janssen, Reuter Foundation, Woodruff Foundation, Reinberger Foundation, National Institute of Health (NIH), and the Centers for Disease Control and Prevention (CDC). Dr. Sajatovic is a consultant to Bracket, Otsuka, Supernus, Neurocrine, Health Analytics and Sunovion and has received royalties from Springer Press, Johns Hopkins University Press, Oxford Press, and UpToDate. Dr. Tatsuoka has research grants from the National Science Foundation, Biogen, and Philips Healthcare. Dr. Lhatoo has research grants from NIH and within the past three years has been a speaker for Sunovion. Authors HL, ECZ, EFC, MEA, KAC, MK, PC, DE have nothing to disclose.
Figures
References
-
- Chong J, Kudrimoti HS, Lopez DCW, Labiner DM. Behavioral risk factors among Arizonans with epilepsy: Behavioral Risk Factor Surveillance System 2005/2006. Epilepsy & Behavior 2010;17: 511–519. - PubMed
-
- Hesdorffer DC, Begley CE. Surveillance of epilepsy and prevention of epilepsy and its sequelae: lessons from the Institute of Medicine report. Current Opinion in Neurology 2013;26: 168–173. - PubMed
-
- Ferguson PL, Chiprich J, Smith G, Dong B, Wannamaker BB, Kobau R, Thurman DJ, Selassie AW. Prevalence of self-reported epilepsy, health care access, and health behaviors among adults in South Carolina. Epilepsy & Behavior 2008;13: 529–534. - PubMed
-
- Begley C, Basu R, Lairson D, Reynolds T, Dubinsky S, Newmark M, Barnwell F, Hauser A, Hesdorffer D. Socioeconomic status, health care use, and outcomes: Persistence of disparities over time. Epilepsia 2011;52: 957–964. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous