Autism-associated Nf1 deficiency disrupts corticocortical and corticostriatal functional connectivity in human and mouse
- PMID: 31128207
- PMCID: PMC6689441
- DOI: 10.1016/j.nbd.2019.104479
Autism-associated Nf1 deficiency disrupts corticocortical and corticostriatal functional connectivity in human and mouse
Abstract
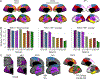
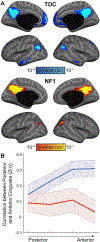
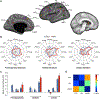
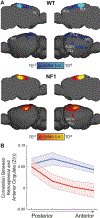
Children with the autosomal dominant single gene disorder, neurofibromatosis type 1 (NF1), display multiple structural and functional changes in the central nervous system, resulting in neuropsychological cognitive abnormalities. Here we assessed the pathological functional organization that may underlie the behavioral impairments in NF1 using resting-state functional connectivity MRI. Coherent spontaneous fluctuations in the fMRI signal across the entire brain were used to interrogate the pattern of functional organization of corticocortical and corticostriatal networks in both NF1 pediatric patients and mice with a heterozygous mutation in the Nf1 gene (Nf1+/-). Children with NF1 demonstrated abnormal organization of cortical association networks and altered posterior-anterior functional connectivity in the default network. Examining the contribution of the striatum revealed that corticostriatal functional connectivity was altered. NF1 children demonstrated reduced functional connectivity between striatum and the frontoparietal network and increased striatal functional connectivity with the limbic network. Awake passive mouse functional connectivity MRI in Nf1+/- mice similarly revealed reduced posterior-anterior connectivity along the cingulate cortex as well as disrupted corticostriatal connectivity. The striatum of Nf1+/- mice showed increased functional connectivity to somatomotor and frontal cortices and decreased functional connectivity to the auditory cortex. Collectively, these results demonstrate similar alterations across species, suggesting that NF1 pathogenesis is linked to striatal dysfunction and disrupted corticocortical connectivity in the default network.
Keywords: ADHD; Autism; Mouse model; Neurofibromatosis type 1; Pediatric patients; fMRI.
Copyright © 2019 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Conflict of interests
The authors declare no competing financial interests.
Figures


Similar articles
-
Functional Connectivity Changes and Executive and Social Problems in Neurofibromatosis Type I.Brain Connect. 2015 Jun;5(5):312-20. doi: 10.1089/brain.2014.0334. Epub 2015 Mar 31. Brain Connect. 2015. PMID: 25705926 Free PMC article.
-
Reduced higher dimensional temporal dynamism in neurofibromatosis type 1.Neuroimage Clin. 2019;22:101692. doi: 10.1016/j.nicl.2019.101692. Epub 2019 Jan 29. Neuroimage Clin. 2019. PMID: 30710873 Free PMC article.
-
Atypical connectivity in the cortico-striatal network in NF1 children and its relationship with procedural perceptual-motor learning and motor skills.J Neurodev Disord. 2022 Mar 1;14(1):15. doi: 10.1186/s11689-022-09428-y. J Neurodev Disord. 2022. PMID: 35232382 Free PMC article.
-
Atypical prefrontal connectivity in attention-deficit/hyperactivity disorder: pathway to disease or pathological end point?Biol Psychiatry. 2011 Jun 15;69(12):1168-77. doi: 10.1016/j.biopsych.2011.03.022. Epub 2011 May 5. Biol Psychiatry. 2011. PMID: 21546000 Review.
-
An update on the central nervous system manifestations of neurofibromatosis type 1.Acta Neuropathol. 2020 Apr;139(4):625-641. doi: 10.1007/s00401-019-02002-2. Epub 2019 Apr 8. Acta Neuropathol. 2020. PMID: 30963251 Free PMC article. Review.
Cited by
-
Polysomnographic study in pediatric neurofibromatosis type 1.Front Neurol. 2023 Jul 18;14:1213430. doi: 10.3389/fneur.2023.1213430. eCollection 2023. Front Neurol. 2023. PMID: 37538252 Free PMC article.
-
Neurofibromatosis Type 1 Implicates Ras Pathways in the Genetic Architecture of Neurodevelopmental Disorders.Behav Genet. 2020 Jul;50(4):191-202. doi: 10.1007/s10519-020-09991-x. Epub 2020 Feb 5. Behav Genet. 2020. PMID: 32026187 Review.
-
Loss of NF1 in Drosophila Larvae Causes Tactile Hypersensitivity and Impaired Synaptic Transmission at the Neuromuscular Junction.J Neurosci. 2022 Dec 14;42(50):9450-9472. doi: 10.1523/JNEUROSCI.0562-22.2022. Epub 2022 Nov 7. J Neurosci. 2022. PMID: 36344265 Free PMC article.
-
Auditory processing in rodent models of autism: a systematic review.J Neurodev Disord. 2022 Aug 30;14(1):48. doi: 10.1186/s11689-022-09458-6. J Neurodev Disord. 2022. PMID: 36042393 Free PMC article.
-
Brain-wide structural and functional disruption in mice with oligodendrocyte-specific Nf1 deletion is rescued by inhibition of nitric oxide synthase.Proc Natl Acad Sci U S A. 2020 Sep 8;117(36):22506-22513. doi: 10.1073/pnas.2008391117. Epub 2020 Aug 24. Proc Natl Acad Sci U S A. 2020. PMID: 32839340 Free PMC article.
References
-
- Bertero A, Liska A, Pagani M, Parolisi R, Masferrer ME, Gritti M, Pedrazzoli M, Galbusera A, Sarica A, Cerasa A, Buffelli M, Tonini R, Buffo A, Gross C, Pasqualetti M, Gozzi A, 2018. Autism-associated 16p11.2 microdeletion impairs prefrontal functional connectivity in mouse and human. Brain. 10.1093/brain/awy111. - DOI - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

