Single intranasal immunization with chimpanzee adenovirus-based vaccine induces sustained and protective immunity against MERS-CoV infection
- PMID: 31130102
- PMCID: PMC6542157
- DOI: 10.1080/22221751.2019.1620083
Single intranasal immunization with chimpanzee adenovirus-based vaccine induces sustained and protective immunity against MERS-CoV infection
Abstract
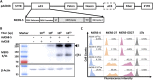
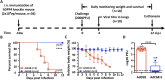
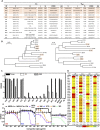
The recently identified Middle East Respiratory Syndrome Coronavirus (MERS-CoV) causes severe and fatal acute respiratory illness in humans. However, no approved prophylactic and therapeutic interventions are currently available. The MERS-CoV envelope spike protein serves as a crucial target for neutralizing antibodies and vaccine development, as it plays a critical role in mediating viral entry through interactions with the cellular receptor, dipeptidyl peptidase 4 (DPP4). Here, we constructed a recombinant rare serotype of the chimpanzee adenovirus 68 (AdC68) that expresses full-length MERS-CoV S protein (AdC68-S). Single intranasal immunization with AdC68-S induced robust and sustained neutralizing antibody and T cell responses in BALB/c mice. In a human DPP4 knock-in (hDPP4-KI) mouse model, it completely protected against lethal challenge with a mouse-adapted MERS-CoV (MERS-CoV-MA). Passive transfer of immune sera to naïve hDPP4-KI mice also provided survival advantages from lethal MERS-CoV-MA challenge. Analysis of sera absorption and isolated monoclonal antibodies from immunized mice demonstrated that the potent and broad neutralizing activity was largely attributed to antibodies targeting the receptor binding domain (RBD) of the S protein. These results show that AdC68-S can induce protective immune responses in mice and represent a promising candidate for further development against MERS-CoV infection in both dromedaries and humans.
Keywords: MERS-CoV vaccine; chimpanzee adenoviral vector; intranasal immunization; monoclonal antibody; receptor binding domain (RBD).
Figures


Similar articles
-
Antibodies and vaccines against Middle East respiratory syndrome coronavirus.Emerg Microbes Infect. 2019;8(1):841-856. doi: 10.1080/22221751.2019.1624482. Emerg Microbes Infect. 2019. PMID: 31169078 Free PMC article. Review.
-
Single-Dose, Intranasal Immunization with Recombinant Parainfluenza Virus 5 Expressing Middle East Respiratory Syndrome Coronavirus (MERS-CoV) Spike Protein Protects Mice from Fatal MERS-CoV Infection.mBio. 2020 Apr 7;11(2):e00554-20. doi: 10.1128/mBio.00554-20. mBio. 2020. PMID: 32265331 Free PMC article.
-
Superior immune responses induced by intranasal immunization with recombinant adenovirus-based vaccine expressing full-length Spike protein of Middle East respiratory syndrome coronavirus.PLoS One. 2019 Jul 22;14(7):e0220196. doi: 10.1371/journal.pone.0220196. eCollection 2019. PLoS One. 2019. PMID: 31329652 Free PMC article.
-
The recombinant N-terminal domain of spike proteins is a potential vaccine against Middle East respiratory syndrome coronavirus (MERS-CoV) infection.Vaccine. 2017 Jan 3;35(1):10-18. doi: 10.1016/j.vaccine.2016.11.064. Epub 2016 Nov 26. Vaccine. 2017. PMID: 27899228 Free PMC article.
-
Prospects for a MERS-CoV spike vaccine.Expert Rev Vaccines. 2018 Aug;17(8):677-686. doi: 10.1080/14760584.2018.1506702. Epub 2018 Aug 9. Expert Rev Vaccines. 2018. PMID: 30058403 Free PMC article. Review.
Cited by
-
Lessons for COVID-19 Immunity from Other Coronavirus Infections.Immunity. 2020 Aug 18;53(2):248-263. doi: 10.1016/j.immuni.2020.07.005. Epub 2020 Jul 14. Immunity. 2020. PMID: 32717182 Free PMC article. Review.
-
A three-antigen Plasmodium falciparum DNA prime-Adenovirus boost malaria vaccine regimen is superior to a two-antigen regimen and protects against controlled human malaria infection in healthy malaria-naïve adults.PLoS One. 2021 Sep 8;16(9):e0256980. doi: 10.1371/journal.pone.0256980. eCollection 2021. PLoS One. 2021. PMID: 34495988 Free PMC article. Clinical Trial.
-
Antibodies and vaccines against Middle East respiratory syndrome coronavirus.Emerg Microbes Infect. 2019;8(1):841-856. doi: 10.1080/22221751.2019.1624482. Emerg Microbes Infect. 2019. PMID: 31169078 Free PMC article. Review.
-
The use of viral vectors in vaccine development.NPJ Vaccines. 2022 Jul 4;7(1):75. doi: 10.1038/s41541-022-00503-y. NPJ Vaccines. 2022. PMID: 35787629 Free PMC article. Review.
-
Immunology of COVID-19: Current State of the Science.Immunity. 2020 Jun 16;52(6):910-941. doi: 10.1016/j.immuni.2020.05.002. Epub 2020 May 6. Immunity. 2020. PMID: 32505227 Free PMC article. Review.
References
-
- WHO Middle East respiratory syndrome coronavirus (MERS-CoV); 2019 http://www.who.int/emergencies/mers-cov/en/</authors. - PubMed
-
- WHO Middle East respiratory syndrome coronavirus (MERS-CoV); 2017 http://www.who.int/emergencies/mers-cov/en/</authors. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
