O-GlcNAc Transferase Promotes Compensated Cardiac Function and Protein Kinase A O-GlcNAcylation During Early and Established Pathological Hypertrophy From Pressure Overload
- PMID: 31131693
- PMCID: PMC6585351
- DOI: 10.1161/JAHA.118.011260
O-GlcNAc Transferase Promotes Compensated Cardiac Function and Protein Kinase A O-GlcNAcylation During Early and Established Pathological Hypertrophy From Pressure Overload
Abstract
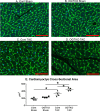
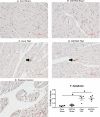
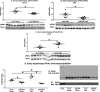
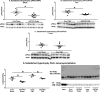
Background Protein posttranslational modifications by O-linked β-N-acetylglucosamine (O-GlcNAc) increase with cardiac hypertrophy, yet the functional effects of these changes are incompletely understood. In other organs, O-GlcNAc promotes adaptation to acute physiological stressors; however, prolonged O-GlcNAc elevations are believed to be detrimental. We hypothesize that early O-GlcNAcylation improves cardiac function during initial response to pressure overload hypertrophy, but that sustained elevations during established pathological hypertrophy negatively impact cardiac function by adversely affecting calcium handling proteins. Methods and Results Transverse aortic constriction or sham surgeries were performed on littermate controls or cardiac-specific, inducible O-GlcNAc transferase knockout (OGTKO) mice to reduce O-GlcNAc levels. O-GlcNAc transferase deficiency was induced at different times. To evaluate the initial response to pressure overload, OGTKO was completed preoperatively and mice were followed for 2 weeks post-surgery. To assess prolonged O-GlcNAcylation during established hypertrophy, OGTKO was performed starting 18 days after surgery and mice were followed until 6 weeks post-surgery. In both groups, OGTKO with transverse aortic constriction caused significant left ventricular dysfunction. OGTKO did not affect levels of the calcium handling protein SERCA2a. OGTKO reduced phosphorylation of phospholamban and cardiac troponin I, which would negatively impact cardiac function. O-GlcNAcylation of protein kinase A catalytic subunit, a kinase for phospholamban, decreased with OGTKO. Conclusions O-GlcNAcylation promotes compensated cardiac function in both early and established pathological hypertrophy. We identified a novel O-GlcNAcylation of protein kinase A catalytic subunit, which may regulate calcium handling and cardiac function.
Keywords: OGT; O‐GlcNAc; cardiac failure; cardiac hypertrophy; pressure overload; protein kinase A phosphorylation.
Figures





Similar articles
-
Fine-tuning the cardiac O-GlcNAcylation regulatory enzymes governs the functional and structural phenotype of the diabetic heart.Cardiovasc Res. 2022 Jan 7;118(1):212-225. doi: 10.1093/cvr/cvab043. Cardiovasc Res. 2022. PMID: 33576380
-
Deficiency in nucleoside diphosphate kinase B leads to endothelial activation of the hexosamine biosynthesis pathway and cardiac dysfunction.Cardiovasc Diabetol. 2025 Feb 21;24(1):84. doi: 10.1186/s12933-025-02633-8. Cardiovasc Diabetol. 2025. PMID: 39985023 Free PMC article.
-
Cardiac O-GlcNAc signaling is increased in hypertrophy and heart failure.Physiol Genomics. 2012 Feb 1;44(2):162-72. doi: 10.1152/physiolgenomics.00016.2011. Epub 2011 Nov 29. Physiol Genomics. 2012. PMID: 22128088
-
Regulation of cardiac O-GlcNAcylation: More than just nutrient availability.Biochim Biophys Acta Mol Basis Dis. 2020 May 1;1866(5):165712. doi: 10.1016/j.bbadis.2020.165712. Epub 2020 Jan 31. Biochim Biophys Acta Mol Basis Dis. 2020. PMID: 32014551 Free PMC article. Review.
-
Foetal recapitulation of nutrient surplus signalling by O-GlcNAcylation and the failing heart.Eur J Heart Fail. 2023 Aug;25(8):1199-1212. doi: 10.1002/ejhf.2972. Epub 2023 Jul 20. Eur J Heart Fail. 2023. PMID: 37434410 Review.
Cited by
-
Role of O-Linked N-Acetylglucosamine Protein Modification in Cellular (Patho)Physiology.Physiol Rev. 2021 Apr 1;101(2):427-493. doi: 10.1152/physrev.00043.2019. Epub 2020 Jul 30. Physiol Rev. 2021. PMID: 32730113 Free PMC article. Review.
-
Best practices in assessing cardiac protein O-GlcNAcylation by immunoblot.Am J Physiol Heart Circ Physiol. 2023 Oct 1;325(4):H601-H616. doi: 10.1152/ajpheart.00104.2023. Epub 2023 Aug 4. Am J Physiol Heart Circ Physiol. 2023. PMID: 37539459 Free PMC article.
-
Protein glycosylation in cardiovascular health and disease.Nat Rev Cardiol. 2024 Aug;21(8):525-544. doi: 10.1038/s41569-024-00998-z. Epub 2024 Mar 18. Nat Rev Cardiol. 2024. PMID: 38499867 Review.
-
Sex alters thyroid hormone's effect on protein O-GlcNAcylation in the aged mouse heart.BMC Mol Cell Biol. 2025 Jun 10;26(1):19. doi: 10.1186/s12860-025-00543-x. BMC Mol Cell Biol. 2025. PMID: 40495125 Free PMC article.
-
New Insights Into the Biology of Protein O-GlcNAcylation: Approaches and Observations.Front Aging. 2021 Mar 12;1:620382. doi: 10.3389/fragi.2020.620382. eCollection 2020. Front Aging. 2021. PMID: 35822169 Free PMC article.
References
-
- Torres CR, Hart GW. Topography and polypeptide distribution of terminal N‐acetylglucosamine residues on the surfaces of intact lymphocytes. Evidence for O‐linked GlcNAc. J Biol Chem. 1984;259:3308–3317. - PubMed
-
- Hu P, Shimoji S, Hart GW. Site‐specific interplay between O‐GlcNAcylation and phosphorylation in cellular regulation. FEBS Lett. 2010;584:2526–2538. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

