A substrate tagging and two-step enzymatic reaction strategy for large-scale synthesis of 2,7-anhydro-sialic acid
- PMID: 31132641
- PMCID: PMC6660240
- DOI: 10.1016/j.carres.2019.05.002
A substrate tagging and two-step enzymatic reaction strategy for large-scale synthesis of 2,7-anhydro-sialic acid
Abstract
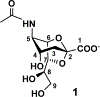
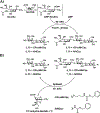
A sialyltransferase acceptor tagging and two-step enzymatic reaction strategy has been developed for multigram-scale chemoenzymatic synthesis of 2,7-anhydro-N-acetylneuraminic acid (2,7-anhydro-Neu5Ac), a compound that can serve as a sole carbon source for the growth of Ruminococcus gnavus, a common human gut commensal. Different approaches of introducing hydrophobic UV-active tags to lactose as well-suited sialyltransferase acceptors have been explored and a simple two-step high-yield chemical synthetic procedure has been identified. The UV-active hydrophobic tag facilitates monitoring reaction progress and allows facile product purification by C18-cartridges. A two-step enzyme-catalyzed reaction procedure has been established to combine with C18 cartridge-based purification process for high-yield production of the desired product in multigram scales with the recycled use of chromophore-tagged lactoside starting material and sialoside intermediate. This study demonstrated an environmentally friendly highly-efficient synthetic and purification strategy for the production of 2,7-anhydro-Neu5Ac to explore its potential functions.
Keywords: 2,7-Anhydro-N-Acetylneuraminic acid; Chemoenzymatic synthesis; Sialic acid; Sialidase; Sialyltransferase; Substrate tagging.
Copyright © 2019 Elsevier Ltd. All rights reserved.
Figures
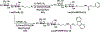

References
-
- Lifely MR, Cottee FH, Formation and identification of two novel anhydro compounds obtained by methanolysis of N-acetylneuraminic acid and carboxyl-reduced, meningococcal polysaccharide, Carbohydr. Res 107 (1982) 187–197.
-
- Schauer R, Schroder C, Shukla AK, New techniques for the investigation of structure and metabolism of sialic acids, Adv. Exp. Med. Biol 174 (1984) 75–86. - PubMed
-
- Suzuki M, Suzuki A, Yamakawa T, Matsunaga E, Characterization of 2,7-anhydro-N-acetylneuraminic acid in human wet cerumen, J. Biochem 97 (1985) 509–515. - PubMed
-
- Li YT, Nakagawa H, Ross SA, Hansson GC, Li SC, A novel sialidase which releases 2,7-anhydro-alpha-N-acetylneuraminic acid from sialoglycoconjugates, J. Biol. Chem 265 (1990) 21629–21633. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

