Electrochemical Reduction of CO2 to Formate on Easily Prepared Carbon-Supported Bi Nanoparticles
- PMID: 31141906
- PMCID: PMC6600365
- DOI: 10.3390/molecules24112032
Electrochemical Reduction of CO2 to Formate on Easily Prepared Carbon-Supported Bi Nanoparticles
Abstract
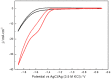
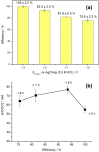
Herein, the electrochemical reduction of CO2 to formate on carbon-supported bismuth nanoparticles is reported. Carbon-supported Bi nanoparticles (about 10 nm in size) were synthesized using a simple, fast and scalable approach performed under room conditions. The so-prepared Bi electrocatalyst was characterized by different physicochemical techniques, including transmission electron microscopy, X-ray photoelectron spectroscopy, and X-ray diffraction and subsequently air-brushed on a carbon paper to prepare electrodes. These electrodes were characterized by scanning electron microscopy, energy-dispersive X-ray spectroscopy and also by cyclic voltammetry. Finally, CO2 electroreduction electrolyses were performed at different electrode potentials for 3 h. At the optimal electrode potential (-1.6 V vs AgCl/Ag), the concentration of formate was about 77 mM with a faradaic efficiency of 93 ± 2.5%. A 100% faradaic efficiency was found at a lower potential (-1.5 V vs AgCl/Ag) with a formate concentration of about 55 mM. In terms of stability, we observed that after about 70 h (in 3 h electrolysis experiments at different potentials), the electrode deactivates due to the gradual loss of metal as shown by SEM/EDX analyses of the deactivated electrodes.
Keywords: Bi electrodes; CO2 reduction; electrocatalysis; formate.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Continuous Electrochemical Reduction of CO2 to Formate: Comparative Study of the Influence of the Electrode Configuration with Sn and Bi-Based Electrocatalysts.Molecules. 2020 Sep 28;25(19):4457. doi: 10.3390/molecules25194457. Molecules. 2020. PMID: 32998373 Free PMC article.
-
Ultrathin Bismuth Nanosheets as a Highly Efficient CO2 Reduction Electrocatalyst.ChemSusChem. 2018 Mar 9;11(5):848-853. doi: 10.1002/cssc.201702229. Epub 2018 Jan 25. ChemSusChem. 2018. PMID: 29323463
-
Rational Design of Sulfur-Doped Copper Catalysts for the Selective Electroreduction of Carbon Dioxide to Formate.ChemSusChem. 2018 Jan 10;11(1):320-326. doi: 10.1002/cssc.201701314. Epub 2017 Oct 12. ChemSusChem. 2018. PMID: 28881436
-
Electrochemical Approaches for CO2 Conversion to Chemicals: A Journey toward Practical Applications.Acc Chem Res. 2022 Mar 1;55(5):638-648. doi: 10.1021/acs.accounts.1c00674. Epub 2022 Jan 18. Acc Chem Res. 2022. PMID: 35041403 Review.
-
Progress in Catalysts for Formic Acid Production by Electrochemical Reduction of Carbon Dioxide.Top Curr Chem (Cham). 2024 Dec 3;383(1):2. doi: 10.1007/s41061-024-00487-4. Top Curr Chem (Cham). 2024. PMID: 39625556 Review.
Cited by
-
Bismuthene for highly efficient carbon dioxide electroreduction reaction.Nat Commun. 2020 Feb 27;11(1):1088. doi: 10.1038/s41467-020-14914-9. Nat Commun. 2020. PMID: 32107389 Free PMC article.
-
Revealing the Intrinsic Restructuring of Bi2O3 Nanoparticles into Bi Nanosheets during Electrochemical CO2 Reduction.ACS Appl Mater Interfaces. 2024 Mar 6;16(9):11552-11560. doi: 10.1021/acsami.3c18285. Epub 2024 Feb 26. ACS Appl Mater Interfaces. 2024. PMID: 38408369 Free PMC article.
-
Efficient electrocatalytic reduction of CO2 on an Ag catalyst in 1-ethyl-3-methylimidazolium ethylsulfate, with its co-catalytic role as a supporting electrolyte during the reduction in an acetonitrile medium.Front Chem. 2025 Apr 9;13:1515903. doi: 10.3389/fchem.2025.1515903. eCollection 2025. Front Chem. 2025. PMID: 40270524 Free PMC article.
-
Continuous Electrochemical Reduction of CO2 to Formate: Comparative Study of the Influence of the Electrode Configuration with Sn and Bi-Based Electrocatalysts.Molecules. 2020 Sep 28;25(19):4457. doi: 10.3390/molecules25194457. Molecules. 2020. PMID: 32998373 Free PMC article.
-
Role of Facets and Morphologies of Different Bismuth-Based Materials for CO2 Reduction to Fuels.Materials (Basel). 2024 Jun 22;17(13):3077. doi: 10.3390/ma17133077. Materials (Basel). 2024. PMID: 38998160 Free PMC article. Review.
References
-
- Figueroa J.D., Fout T., Plasynski S., McIlvried H., Srivastava R.D. Advances in CO2 capture technology—The U.S. Department of Energy’s Carbon Sequestration Program. Int. J. Greenh. Gas Con. 2008;2:9–20. doi: 10.1016/S1750-5836(07)00094-1. - DOI
-
- Qiao J., Liu Y., Zhang J. In: Electrochemical Reduction of Carbon Dioxide: Fundamentals and Technologies. 1st ed. Qiao J., Liu Y., Zhang J., editors. CRC Press; Boca Raton, FL, USA: 2016.
-
- Wu J.J., Zhou X.D. Catalytic conversion of CO2 to value added fuels: Current status, challenges, and future directions. Chin. J. Catal. 2016;37:999–1015. doi: 10.1016/S1872-2067(16)62455-5. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

