The Potential of Stem Cell Therapy to Repair White Matter Injury in Preterm Infants: Lessons Learned From Experimental Models
- PMID: 31143126
- PMCID: PMC6521595
- DOI: 10.3389/fphys.2019.00540
The Potential of Stem Cell Therapy to Repair White Matter Injury in Preterm Infants: Lessons Learned From Experimental Models
Abstract
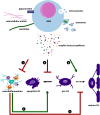
Diffuse white matter injury (dWMI) is a major cause of morbidity in the extremely preterm born infant leading to life-long neurological impairments, including deficits in cognitive, motor, sensory, psychological, and behavioral functioning. At present, no treatment options are clinically available to combat dWMI and therefore exploration of novel strategies is urgently needed. In recent years, the pathophysiology underlying dWMI has slowly started to be unraveled, pointing towards the disturbed maturation of oligodendrocytes (OLs) as a key mechanism. Immature OL precursor cells in the developing brain are believed to be highly sensitive to perinatal inflammation and cerebral oxygen fluctuations, leading to impaired OL differentiation and eventually myelination failure. OL lineage development under normal and pathological circumstances and the process of (re)myelination have been studied extensively over the years, often in the context of other adult and pediatric white matter pathologies such as stroke and multiple sclerosis (MS). Various studies have proposed stem cell-based therapeutic strategies to boost white matter regeneration as a potential strategy against a wide range of neurological diseases. In this review we will discuss experimental studies focusing on mesenchymal stem cell (MSC) therapy to reduce white matter injury (WMI) in multiple adult and neonatal neurological diseases. What lessons have been learned from these previous studies and how can we translate this knowledge to application of MSCs for the injured white matter in the preterm infant? A perspective on the current state of stem cell therapy will be given and we will discuss different important considerations of MSCs including cellular sources, timing of treatment and administration routes. Furthermore, we reflect on optimization strategies that could potentially reinforce stem cell therapy, including preconditioning and genetic engineering of stem cells or using cell-free stem cell products, to optimize cell-based strategy for vulnerable preterm infants in the near future.
Keywords: brain development; cell therapies; mesenchymal stem cells; myelin loss; preterm birth; regeneration; white matter injury; white matter pathology.
Figures

References
Publication types
LinkOut - more resources
Full Text Sources

