Fabrication of biocompatible porous scaffolds based on hydroxyapatite/collagen/chitosan composite for restoration of defected maxillofacial mandible bone
- PMID: 31144260
- PMCID: PMC6825626
- DOI: 10.1007/s40204-019-0113-x
Fabrication of biocompatible porous scaffolds based on hydroxyapatite/collagen/chitosan composite for restoration of defected maxillofacial mandible bone
Abstract
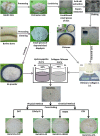
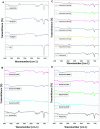
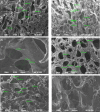
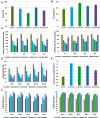
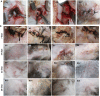
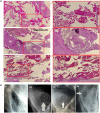
Fabrication of scaffolds from biomaterials for restoration of defected mandible bone has attained increased attention due to limited accessibility of natural bone for grafting. Hydroxyapatite (Ha), collagen type 1 (Col1) and chitosan (Cs) are widely used biomaterials which could be fabricated as a scaffold to overcome the paucity of bone substitutes. Here, rabbit Col1, shrimp Cs and bovine Ha were extracted and characterized with respect to physicochemical properties. Following the biocompatibility, degradability and cytotoxicity tests for Ha, Col1 and Cs a hydroxyapatite/collagen/chitosan (Ha·Col1·Cs) scaffold was fabricated using thermally induced phase separation technique. This scaffold was cross-linked with (1) either glutaraldehyde (GTA), (2) de-hydrothermal treatment (DTH), (3) irradiation (IR) and (4) 2-hydroxyethyl methacrylate (HEMA), resulting in four independent types (Ha·Col1·Cs-GTA, Ha·Col1·Cs-IR, Ha·Col1·Cs-DTH and Ha·Col1·Cs-HEMA). The developed composite scaffolds were porous with 3D interconnected fiber microstructure. However, Ha·Col1·Cs-IR and Ha·Col1·Cs-GTA showed better hydrophilicity and biodegradability. All four scaffolds showed desirable blood biocompatibility without cytotoxicity for brine shrimp. In vitro studies in the presence of human amniotic fluid-derived mesenchymal stem cells revealed that Ha·Col1·Cs-IR and Ha·Col1·Cs-DHT scaffolds were non-cytotoxic and compatible for cell attachment, growth and mineralization. Further, grafting of Ha·Col1·Cs-IR and Ha·Col1·Cs-DHT was performed in a surgically created non-load-bearing rabbit maxillofacial mandible defect model. Histological and radiological observations indicated the restoration of defected bone. Ha·Col1·Cs-IR and Ha·Col1·Cs-DHT could be used as an alternative treatment in bone defects and may contribute to further development of scaffolds for bone tissue engineering.
Keywords: AF-MSCs; Biocompatibility; Bone tissue engineering; Chitosan; Collagen; Hydroxyapatite; Mandible bone defect; Scaffold.
Conflict of interest statement
The authors declare that they have no competing interest.
Figures

Similar articles
-
Full physicochemical and biocompatibility characterization of a supercritical CO2 sterilized nano-hydroxyapatite/chitosan biodegradable scaffold for periodontal bone regeneration.Biomater Adv. 2023 Mar;146:213280. doi: 10.1016/j.bioadv.2023.213280. Epub 2023 Jan 7. Biomater Adv. 2023. PMID: 36682201
-
Zero-order controlled release of BMP2-derived peptide P24 from the chitosan scaffold by chemical grafting modification technique for promotion of osteogenesis in vitro and enhancement of bone repair in vivo.Theranostics. 2017 Feb 27;7(5):1072-1087. doi: 10.7150/thno.18193. eCollection 2017. Theranostics. 2017. PMID: 28435449 Free PMC article.
-
Anti-infective efficacy, cytocompatibility and biocompatibility of a 3D-printed osteoconductive composite scaffold functionalized with quaternized chitosan.Acta Biomater. 2016 Dec;46:112-128. doi: 10.1016/j.actbio.2016.09.035. Epub 2016 Sep 26. Acta Biomater. 2016. PMID: 27686039
-
Manufacturing methods, properties, and potential applications in bone tissue regeneration of hydroxyapatite-chitosan biocomposites: A review.Int J Biol Macromol. 2023 Jul 15;243:125150. doi: 10.1016/j.ijbiomac.2023.125150. Epub 2023 Jun 5. Int J Biol Macromol. 2023. PMID: 37285882 Review.
-
Nanomaterials for Periodontal Tissue Engineering: Chitosan-Based Scaffolds. A Systematic Review.Nanomaterials (Basel). 2020 Mar 25;10(4):605. doi: 10.3390/nano10040605. Nanomaterials (Basel). 2020. PMID: 32218206 Free PMC article. Review.
Cited by
-
Multislice CT-guided evaluation of collagen-chitosan composite in promoting antebrachiocarpal arthrodesis in a rabbit model.Ir Vet J. 2025 Aug 26;78(1):18. doi: 10.1186/s13620-025-00307-1. Ir Vet J. 2025. PMID: 40859361
-
Biocompatibility Studies on a Collagen-Hydroxyapatite Biomaterial.Curr Health Sci J. 2022 Apr-Jun;48(2):217-225. doi: 10.12865/CHSJ.48.02.12. Epub 2022 Jun 30. Curr Health Sci J. 2022. PMID: 36320879 Free PMC article.
-
Antibacterial Activity of Ulva/Nanocellulose and Ulva/Ag/Cellulose Nanocomposites and Both Blended with Fluoride against Bacteria Causing Dental Decay.Polymers (Basel). 2023 Feb 20;15(4):1047. doi: 10.3390/polym15041047. Polymers (Basel). 2023. PMID: 36850336 Free PMC article.
-
UV-assisted synthesis of hydroxyapatite from eggshells at ambient temperature: cytotoxicity, drug delivery and bioactivity.RSC Adv. 2021 Jan 19;11(6):3686-3694. doi: 10.1039/d0ra09673c. eCollection 2021 Jan 14. RSC Adv. 2021. PMID: 35424272 Free PMC article.
-
Chitosan-collagen-hydroxyapatite membranes for tissue engineering.J Mater Sci Mater Med. 2022 Jan 24;33(2):18. doi: 10.1007/s10856-022-06643-w. J Mater Sci Mater Med. 2022. PMID: 35072812 Free PMC article.
References
-
- Alaribe FN, Manoto SL, Motaung SCKM. Scaffolds from biomaterials: advantages and limitations in bone and tissue engineering. Biologia. 2016;71(4):353–366. doi: 10.1515/biolog-2016-0056. - DOI
-
- Calabrese G, Giuffrida R, Forte S, Fabbi C, Figallo E, Salvatorelli L, Memeo L, Parenti R, Gulisano M, Gulino R. Human adipose-derived mesenchymal stem cells seeded into a collagen-hydroxyapatite scaffold promote bone augmentation after implantation in the mouse. Sci Rep. 2017;7:7110. doi: 10.1038/s41598-017-07672-0. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

