In vitro aged, hiPSC-origin engineered heart tissue models with age-dependent functional deterioration to study myocardial infarction
- PMID: 31146032
- PMCID: PMC6779061
- DOI: 10.1016/j.actbio.2019.05.064
In vitro aged, hiPSC-origin engineered heart tissue models with age-dependent functional deterioration to study myocardial infarction
Abstract
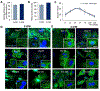
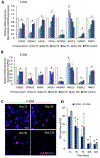
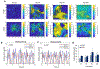
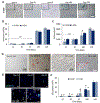
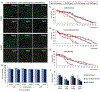
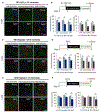
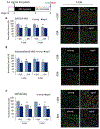
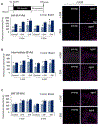
Deaths attributed to ischemic heart disease increased by 41.7% from 1990 to 2013. This is primarily due to an increase in the aged population, however, research on cardiovascular disease (CVD) has been overlooking aging, a well-documented contributor to CVD. The use of young animals is heavily preferred due to lower costs and ready availability, despite the prominent differences between young and aged heart structure and function. Here we present the first human induced pluripotent stem cell (hiPSC)-derived cardiomyocyte (iCM)-based, in vitro aged myocardial tissue model as an alternative research platform. Within 4 months, iCMs go through accelerated senescence and show cellular characteristics of aging. Furthermore, the model tissues fabricated using aged iCMs, with stiffness resembling that of aged human heart, show functional and pharmacological deterioration specific to aged myocardium. Our novel tissue model with age-appropriate physiology and pathology presents a promising new platform for investigating CVD or other age-related diseases. STATEMENT OF SIGNIFICANCE: In vitro and in vivo models of cardiovascular disease are aimed to provide crucial insight on the pathology and treatment of these diseases. However, the contribution of age-dependent cardiovascular changes is greatly underestimated through the use of young animals and premature cardiomyocytes. Here, we developed in vitro aged cardiac tissue models that mimic the aged heart tissue microenvironment and cellular phenotype and present the first evidence that age-appropriate in vitro disease models can be developed to gain more physiologically-relevant insight on development, progression, and amelioration of cardiovascular diseases.
Keywords: Aging; Human induced pluripotent stem cell; Reperfusion injury; Tissue engineering.
Copyright © 2019 Acta Materialia Inc. Published by Elsevier Ltd. All rights reserved.
Conflict of interest statement
7. Disclosure
Authors report no competing financial interests
Figures

References
-
- A.H. Association, Heart disease, stroke and research statistics at-a-glance, AHA Website. (2016) 1–5. doi:10.1007/s13398-014-0173-7.2. - DOI
-
- Anversa P, Palackal T, Sonnenblick EH, Olivetti G, Meggs LG, Capasso JM, Myocyte cell loss and myocyte cellular hyperplasia in the hypertrophied aging rat heart, Circ. Res 67 (1990) 871–885. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

